Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- Published: 13 September 2021

Malaria: a problem to be solved and a time to be bold
- Pedro L. Alonso 1
Nature Medicine volume 27 , pages 1506–1509 ( 2021 ) Cite this article
8453 Accesses
15 Citations
147 Altmetric
Metrics details
Vaccines alone will not be sufficient for the eradication of malaria, which will also require investment in health professionals, better use of data, and universal access to quality health services.
COVID-19 has provided a wake-up call that infectious diseases can have huge health, economic and social costs and require investments that protect the wellbeing of people across the world. By threatening the health and economies of all countries, unprecedented financial efforts and incentives were deployed in a very short period of time to develop and implement new tools against COVID-19, especially vaccines.

This unprecedented investment has revealed the extraordinary power of science, delivering several safe and effective vaccines within months of the sequence of SARS-CoV-2 being determined and shared. The investment and benefits from science, coupled with global solidarity and a focus on equity, should ensure sufficient supply and the equitable distribution of vaccines. It should also be extended to other diseases, such as malaria, that currently threaten the lives of millions of people around the world.
A golden era
History demonstrates that such an investment in malaria can reap remarkable benefits. At the start of the twenty-first century, the transmission of malaria was taking place in 107 countries across five continents, where more than 80% of the world’s population lived 1 . Sub-Saharan Africa carried the brunt of the disease, where a child probably died of malaria every 45 seconds and efforts to control the disease were very limited. Yet this desperate situation had not gone unnoticed.
Towards the end of the last century, there was a growing political and scientific momentum that would lead to a golden era in the fight against this ancient killer of humankind. Insecticide-treated bed nets, artemisinin-based combination therapies, rapid diagnostic tests, new approaches to chemoprevention of target groups with drugs (such as intermittent preventive treatment in pregnancy or in infants, and seasonal malaria chemoprevention) were the extraordinary outputs of a small and underfunded research community.
On the political front, the Abuja Declaration by African Heads of State, followed by the inclusion of malaria in the United Nations Millennium Development Goals, was accompanied by the creation of new funding mechanisms such as The Global Fund to Fight AIDS, Tuberculosis and Malaria, and the US President’s Malaria Initiative 2 , 3 , 4 , 5 . For the first time, substantial financial resources ensured that antimalarial tools could be procured and delivered at scale. The effect has been staggering: 7.6 million deaths and 1.5 billion cases averted in the first 20 years of this century 1 .
These gains have also occurred at a time when many malaria endemic countries have experienced considerable economic growth and social development, both of which are important contributors to decreasing malaria burden. In sub-Saharan Africa, GDP has grown by an average of 4% per annum in the period from 2000 to 2019 6 . During the same time, access to electricity has increased threefold 7 and the percentage of the African population in urban settings has increased from 31% to 41% (ref. 8 ). It is the combined efforts from the scientific community, leadership across the world and socioeconomic development that have contributed to a public health success story and a great return on global health investment.
However, the past five years have shown both the success and the limitations of this effort 1 . More than half (46) of the 87 malaria endemic countries in 2019 are within reach of eliminating the transmission of malaria within their borders. These countries, mostly outside Africa, now account for less than 0.2% of all malaria cases globally, and some, such as El Salvador and China, have recently been certified malaria free 1 , 9 , 10 .
However, for a considerable proportion of the remaining countries with ongoing malaria transmission, reductions in disease incidence and mortality rates have slowed down, particularly in sub-Saharan Africa, where 94% of global malaria cases and deaths occur.
Funding, from both international partners and the endemic countries themselves, has stalled. In the face of a doubling of the population over the past 20 years in sub-Saharan Africa alone, the stark reality is that after US$26 billion of investment to tackle malaria in this region, the estimated number of malaria cases are slightly higher in 2019 (215 million) than in 2000 (204 million) 1 .
The world is therefore likely to continue to see success as a good number of countries become malaria free. However, we are not on track to achieve the agreed targets for reductions in morbidity and mortality by 2030 as set out in the WHO Global Technical Strategy 11 , and malaria eradication is not within sight. In 2017, the WHO raised the alarm on the stalling of progress by declaring that the world was at a crossroads in the fight against malaria 12 , 13 .
Sustaining the gains
First, more financial resources are required. Financing mechanisms and governance also need to acknowledge and enable the leadership of malaria endemic countries, who in turn need to take greater financial responsibility.
Second, plans and activities to control malaria have to be imbedded in the Universal Health Coverage and Primary Health Care agenda 14 , 15 . Robust, resilient, quality health systems are essential to tackle malaria.
Third, data can also support subnational operations, inspired by a problem-solving mindset, to move away from a one-size fits all approach. Countries must invest in quality health-management information systems. The scale up of point-of-care malaria diagnosis, the gradual switch to electronic databases and the investment in malariometric surveys in recent years, which determine the level of malaria in specific locations, have led to increasing availability of reasonable quality data 1 .
Geospatial and mathematical modeling approaches can take into account the inherent heterogeneity of malaria transmission as well as contextual elements, such as access to health facilities, urbanization, important social determinants and other factors. This allows stratification at a national and subnational level, empowering governments to reliably define the optimal mix of malaria interventions that will achieve maximum effect within a given resource envelope.
This sub-nationally tailored approach must extend beyond what is delivered to include local decisions of how to efficiently and equitably deliver to those in need. A sub-national approach will also require further investments in improving surveillance systems, establishment of dynamic integrated national repositories to systematically curate relevant data and national capacity to analyze and make use of the data for locally tailored responses.
Fourth, the malaria community needs to acknowledge the strength and limitations of the tools and strategies available today. We should be able to diagnose and treat all malaria cases. Affordable and easy to use point-of-care diagnostics allow a parasitological confirmatory test. Similarly, safe and highly effective antimalarial medicines exist and can be used to treat all infections. Consequently, no one should be dying of malaria. However, more than 400,000 malaria deaths continue to take place every year owing to a lack of access to prompt quality care.
Preventable deaths
Prevention relies on both tools against the anopheline vector, such as insecticide-treated mosquito nets, and the use of medicines to prevent infections in key target groups. When properly implemented, prevention with medicine results in a substantial reduction in disease and malaria deaths.
However, a lack of prioritization in research and development means that there is a lack of optimal drugs, regimes and formulations for prevention, which represent a barrier to adoption and impact. Furthermore, both drugs and diagnostics will remain challenged by the emergence and spread of drug resistance or parasite gene deletions, a threat towards which the malaria community has so far responded effectively.
In most of sub-Saharan Africa, long-lasting insecticide-treated nets (LLINs) represent the cornerstone of efforts to control the malaria vector. The efficacy of LLINs in the prevention of disease is modest—around 45% in children under the age of five 16 —and the nets need to be replaced every three years. Part of the efficacy of LLINs relies on impregnation with insecticide, against which Anopheline mosquitoes can develop resistance, and they also rely on quality and integrity, which may represent an even greater challenge 17 .
New prevention tools are needed, including against the mosquito vector and antimalarial medicines. Monoclonal antibodies may also provide opportunities to prevent infection for several months during the periods of highest risk for the key target populations.
Various vaccines
COVID-19 reminds us that vaccines remain the most important tool to prevent communicable diseases. The quest for a malaria vaccine started more than a century ago. Today, there is a first-generation malaria vaccine (RTS,S produced by GSK) based on a recombinant protein that targets the circumsporozoite protein, the predominant sporozoite surface protein of Plasmodium falciparum . RTS,S has completed its clinical development and received a positive scientific opinion by the European Medicines Agency 18 . The efficacy of RTS,S is modest — around 40% reduction of disease.
RTS,S is currently undergoing large-scale pilot implementation that involves several hundreds of thousands of children in three African countries, and will be considered for a potential WHO recommendation for broader scale use before the end of the year. If this historic decision is reached, it would represent the first time that a vaccine against a human malaria parasite is recommended for public-heath use and a first vaccine incorporated to the antimalarial armamentarium toolbox, with the potential to avert millions of cases and hundreds of thousands of deaths. More importantly, this first-generation vaccine will show that the 30-year-long development effort has yielded a safe and effective vaccine against a complex parasite.
A limited number of other vaccine candidates are being tested. One of the most advanced is the R21/MM vaccine candidate. This is based on a similar circumsporozoite antigen to RTS,S and has been developed using the Matrix-M adjuvant platform. In a phase 2b trial, R21/MM showed 77% protection over a one-year follow up, among 450 children living in an area of intensely seasonal malaria transmission in Burkina Faso 19 . The results of the phase 2b trial are encouraging, and has increased anticipation for phase 3 trials that, crucially, need to include areas where malaria occurs all year round, as well as long-term follow up to allow adequate comparison with RTS,S.
In recent years, the use of whole parasite immunization strategies has been tried. Whole-cell P. falciparum sporozoite ( Pf SPZ)-based vaccines are a promising way to evoke immunity, as a broad antigenic repertoire of the parasite is present in the pre-erythrocytic development stages, especially in the liver phase. Sporozoites either attenuated by radiation, or with their development arrested via antimalarial medicines, have been reported to induce 100% protection against heterologous challenge in a small number of volunteers 20 .
BioNTech has recently announced its intention to develop a malaria vaccine using its mRNA technology, which has been used so effectively against COVID-19 21 . The goal is to yield a tool that will enable eradication, with a high efficacy in preventing infection and with a long duration of protection. This declaration represents a U-turn in the decades-long trend of major vaccine developers abandoning or scaling down their efforts in the field of malaria. This announcement also represents the potential of new visions and approaches from the field of oncology being applied to address the challenges and complexities of inducing immunity against complex parasites.
Malaria has plagued humans for millennia and has led to an unimaginable loss of life. Malaria has also had an important role in the geopolitics and evolutionary history of humans. The malaria problem is evolving, dynamic and diverse, but it is now concentrated in some of the poorest communities in the world. The lessons of the past two decades show that success in malaria is possible when the world pulls together. However, enormous biological, political, governance, socioeconomic, data and financial challenges remain. These challenges require the world to be bold again.
New tools, including vaccines, could represent game changers, and demand a collective effort that builds on scientific learning and collaboration. But vaccines by themselves will not be a solution to the problem of malaria. No progress will be made without a well-trained and empowered cadre of health workers. Better use of data to plan, implement and track progress, even within a single country, will allow resources to be used more efficiently.
Finally, national decision-making processes must be at the core of public health governance. Governments must identify and serve those communities that are consistently not reached with quality malaria services, especially those delivered through the public health system. Only then can we move again toward global eradication.
World Health Organization. https://www.who.int/publications/i/item/9789240015791 (2020).
World Health Organization. https://apps.who.int/iris/handle/10665/67816 (2000).
United Nations. https://www.un.org/millenniumgoals/ (2015).
The Global Fund. https://www.theglobalfund.org/en/overview/ (2019).
PMI. https://www.pmi.gov/about (2015).
The World Bank. https://data.worldbank.org/indicator/NY.GDP.PCAP.CD (2020).
The World Bank. https://data.worldbank.org/indicator/EG.ELC.ACCS.RU.ZS (2020).
United Nations. https://population.un.org/wpp/ (2019).
PAHO. https://www.paho.org/en/topics/malaria/salvador-first-central-american-country-receive-who-malaria-free-country (2021).
World Health Organization. https://www.who.int/news/item/30-06-2021-from-30-million-cases-to-zero-china-is-certified-malaria-free-by-who (2021).
World Health Organization. https://www.who.int/malaria/areas/global_technical_strategy/en/ (2015).
World Health Organization. https://www.who.int/malaria/publications/world-malaria-report-2017/en/ (2017).
Alonso, P. & Noor, A. M. Lancet 390 , 2532–2534 (2017).
Article Google Scholar
World Health Organization. https://www.who.int/teams/primary-health-care/conference/declaration (2018).
United Nations. https://www.un.org/pga/73/event/universal-health-coverage/ (2019).
Lengeler, C. Cochrane Database Syst. Rev. 2 , CD000363 (2004).
Google Scholar
Lindsay, S. W., Thomas, M. B. & Kleinschmidt, I. Lancet 9 , e1325–e1331 (2016).
World Health Organization. https://www.who.int/immunization/sage/meetings/2015/october/1_Final_malaria_vaccine_background_paper_v2015_09_30.pdf (2015).
Datoo, M. S. et al. Lancet 397 , 1809–1818 (2021).
Article CAS Google Scholar
Mwakingwe-Omari, A. et al. Nature 595 , 289–294 (2021).
Cheney, C. https://www.devex.com/news/plans-for-new-malaria-vaccine-underscore-benefits-of-mrna-beyond-covid-19-100497 (2021).
Download references
Acknowledgements
This manuscript has benefitted from several discussions with partners and colleagues within the Global Malaria Programme, particularly A. Noor, D. Schellenberg and A. Robb.
Author information
Authors and affiliations.
Global Malaria Programme, World Health Organization, Geneva, Switzerland
Pedro L. Alonso
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Pedro L. Alonso .
Ethics declarations
Competing interests.
The author declares no competing interests.
Rights and permissions
Reprints and permissions
About this article
Cite this article.
Alonso, P.L. Malaria: a problem to be solved and a time to be bold. Nat Med 27 , 1506–1509 (2021). https://doi.org/10.1038/s41591-021-01492-6
Download citation
Published : 13 September 2021
Issue Date : September 2021
DOI : https://doi.org/10.1038/s41591-021-01492-6
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
This article is cited by
Optimizing malaria vector control in the greater mekong subregion: a systematic review and mathematical modelling study to identify desirable intervention characteristics.
- Yuqian Wang
- Nakul Chitnis
- Emma L. Fairbanks
Parasites & Vectors (2024)
Indigenous medicinal plants used in folk medicine for malaria treatment in Kwara State, Nigeria: an ethnobotanical study
- Ikponmwosa Owen Evbuomwan
- Oluyomi Stephen Adeyemi
- Olarewaju Michael Oluba
BMC Complementary Medicine and Therapies (2023)
Social representations of malaria in a southern Malian community: an ethnographic qualitative study
- Bourema Sissoko
- Mohamed Yunus Rafiq
- N’bamori dite Naba Sissoko
Malaria Journal (2022)
Dynamical malaria modeling as a tool for bold policy-making
- Branwen Nia Owen
- Munir Winkel
- Emilie Pothin
Nature Medicine (2022)
Inferring the epidemiological benefit of indoor vector control interventions against malaria from mosquito data
- Ellie Sherrard-Smith
- Corine Ngufor
- Thomas S. Churcher
Nature Communications (2022)
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
Sign up for the Nature Briefing newsletter — what matters in science, free to your inbox daily.

Malaria: Obstacles and Opportunities (1991)
Chapter: 1. conclusions and recommendations, conclusions and recommendations, defining the problem.
The outlook for malaria control is grim. The disease, caused by mosquito-borne parasites, is present in 102 countries and is responsible for over 100 million clinical cases and 1 to 2 million deaths each year. Over the past two decades, efforts to control malaria have met with less and less success. In many regions where malaria transmission had been almost eliminated, the disease has made a comeback, sometimes surpassing earlier recorded levels. The dream of completely eliminating malaria from many parts of the world, pursued with vigor during the 1950s and 1960s, has gradually faded. Few believe today that a global eradication of malaria will be possible in the foreseeable future.
Worldwide, the number of cases of malaria caused by Plasmodium falciparum , the most dangerous species of the parasite, is on the rise. Drug-resistant strains of P. falciparum are spreading rapidly, and there have been recent reports of drug resistance in people infected with P. vivax , a less virulent form of the parasite. Furthermore, mosquitoes are becoming increasingly resistant to insecticides, and in many cases, have adapted so as to avoid insecticide-treated surfaces altogether.
In large part because of the spread of drug and insecticide resistance, there are fewer tools available today to control malaria than there were 20 years ago. In many countries, the few remaining methods are often ap-
plied inappropriately. The situation in many African nations is particularly dismal, exacerbated by a crumbling health infrastructure that has made the implementation of any disease control program difficult.
Malaria cases among tourists, business travelers, military personnel, and migrant workers in malarious areas have been increasing steadily in the last several years, posing new concerns that the disease will be introduced to currently nonmalarious areas. Recent epidemics have claimed tens of thousands of lives in Africa, and there is an increasing realization that malaria is a major impediment to socioeconomic development in many countries. Unless practical, cost-effective strategies can be developed and successfully implemented, malaria will continue to exact a heavy toll on human life and health around the world.
Although often considered a single disease, malaria is more accurately viewed as many diseases, each shaped by subtle interactions of biologic, ecologic, social, and economic factors. The species of parasite, the behavior of the mosquito host, the individual's immune status, the climate, human activities, and access to health services all play important roles in determining the intensity of disease transmission, who will become infected, who will get sick, and who will die.
Gem miners along the Thailand-Cambodia border, American tourists on a wildlife safari in East Africa, villagers living on the central highlands in Madagascar, residents of San Diego County, California, a young pregnant woman in Malawi, Swiss citizens living near Geneva International Airport, children in Africa south of the Sahara, and a U.S. State Department secretary in Tanzania seem to have little in common, yet they are all at risk of contracting malaria. Because of the disease's variable presentations, each will be affected differently, as illustrated below.
For the hundreds of thousands of Thai seasonal agricultural workers who travel deep into the forest along the Thailand-Cambodia border to mine for gems, malaria is the cost of doing business. These young men are exposed to aggressive forest mosquitoes, and within two to three weeks after arriving, almost every miner will get malaria. Many gem miners seek medications to prevent and self-treat mild cases of the disease. But because malaria in this part of the world is resistant to most antimalarial drugs, the few effective drugs are reserved for the treatment of confirmed cases of malaria. To complicate matters, there are no health services in the forest to treat patients, and the health clinics in Thailand are overburdened by the high demand for treating those with severe malaria, most of whom are returning gem miners. A similar scenario involving over 400,000 people exists among gold miners in Rondonia, Brazil.
Each year, over seven million U.S. citizens visit parts of the world
where malaria is present. Many, at the recommendation of their travel agent or physician, take antimalarial medications as a preventive measure, but a significant number do not. Tourists and other travelers who have never been exposed to malaria, and therefore have never developed protective immunity, are at great risk for contracting severe disease. Ironically, it is not the infection itself that poses the biggest danger, but the chance that treatment will be delayed because of misdiagnosis upon the individual's return to the United States. Most U.S. doctors have never seen a patient with malaria, are often confused by the wide array of symptoms, and are largely unaware that malaria in a nonimmune person can be a medical emergency, sometimes rapidly fatal.
Prior to 1950, malaria was the major cause of death in the central highlands of the African island nation of Madagascar. In the late 1950s, an aggressive program of indoor insecticide spraying rid the area of malaria-carrying mosquitoes, and malaria virtually disappeared. By the 1970s, confident of a victory in the battle against malaria, Madagascar began to phase out its spraying program; in some areas spraying was halted altogether. In the early 1980s, the vector mosquitoes reinvaded the central highlands, and in 1986 a series of devastating epidemics began. The older members of the population had long since lost the partial immunity they once had, and the younger island residents had no immunity at all. During the worst of the epidemics, tens of thousands of people died in one three-month period. The tragedy of this story is that it could have been prevented. A cheap antimalarial drug, chloroquine, could have been a powerful weapon in Madagascar, where drug resistance was not a significant concern. Because of problems in international and domestic drug supply and delivery, however, many people did not receive treatment and many died. In the last 18 months, surveillance has improved, spraying against the mosquito has resumed, and more effective drug distribution networks have been established. Malaria-related mortality has declined sharply as a result.
Malaria, once endemic in the southern United States, occurs relatively infrequently. Indeed, there have been only 23 outbreaks of malaria since 1950, and the majority of these occurred in California. But for each of the past three years, the San Diego County Department of Health Services has had to conduct an epidemiologic investigation into local transmission of malaria. An outbreak in the late summer of 1988 involved 30 persons, the largest such outbreak in the United States since 1952. In the summer of 1989, three residents of San Diego County—a migrant worker and two permanent residents—were diagnosed with malaria; in 1990, a teenager living in a suburb of San Diego County fell ill with malaria. All of the cases were treated successfully, but these incidents raise questions about the possibility of new and larger outbreaks in the future. Malaria
transmission in San Diego County (and in much of California) is attributed to the presence of individuals from malaria-endemic regions who lack access to medical care, the poor shelter and sanitation facilities of migrant workers, and the ubiquitous presence of Anopheles mosquitoes in California.
A 24-year-old pregnant Yao woman from the Mangochi District in Malawi visited the village health clinic monthly to receive prenatal care. While waiting to be seen by the health provider, she and other women present listened to health education talks which were often about the dangers of malaria during pregnancy, and the need to install screens around the house to keep the mosquitoes away, to sleep under a bednet, and to take a chloroquine tablet once a week. Toward the end of her second trimester of pregnancy, the woman returned home from her prenatal visit with her eight tablets of chloroquine wrapped in a small packet of brown paper. She promptly gave the medicine to her husband to save for the next time he or one of their children fell ill. The next week she developed a very high malarial fever and went into labor prematurely. The six-month-old fetus was born dead.
Over a two-week period in the summer of 1989, five Swiss citizens living within a mile of Geneva International Airport presented at several hospitals with acute fever and chills. All had malaria. Four of the five had no history of travel to a malarious region; none had a history of intravenous drug use or blood transfusion. Apart from their symptoms, the only thing linking the five was their proximity to the airport. A subsequent epidemiologic investigation suggested that the malaria miniepidemic was caused by the bite of stowaway mosquitoes en route from a malaria-endemic country. The warm weather, lack of systematic spraying of aircraft, and the close proximity of residential areas to the airport facilitated the transmission of the disease.
Malaria is a part of everyday life in Africa south of the Sahara. Its impact on children is particularly severe. Mothers who bring unconscious children to the hospital often report that the children were playing that morning, convulsed suddenly, and have been unconscious ever since. These children are suffering from the most frequently fatal complication of the disease, cerebral malaria. Other children succumb more slowly to malaria, becoming progressively more anemic with each subsequent infection. By the time they reach the hospital, they are too weak to sit and are literally gasping for breath. Many children are brought to hospitals as a last resort, after treatment given for “fever” at the local health center has proved ineffective. Overall, children with malaria account for a third of all hospital admissions. A third of all children hospitalized for malaria die. In most parts of Africa, there are no effective or affordable options to prevent the
disease, so children are at high risk until they have been infected enough times to develop a partial immunity.
A 52-year-old American woman, the secretary to the U.S. ambassador in Tanzania, had been taking a weekly dose of chloroquine to prevent malaria since her arrival in the country the year before. She arrived at work one morning complaining of exhaustion, a throbbing headache, and fever. A blood sample was taken and microscopically examined for malaria parasites. She was found to be infected with P. falciparum , and was treated immediately with high doses of chloroquine. That night, she developed severe diarrhea, and by morning she was found to be disoriented and irrational. She was diagnosed as having cerebral malaria, and intravenous quinine treatment was started. Her condition gradually deteriorated—she became semicomatose and anemic, and approximately 20 percent of her red blood cells were found to be infected with malaria parasites. After continued treatment for several days, no parasites were detected in her blood. Despite receiving optimal care, other malaria-related complications developed and she died just nine days after the illness began. The cause of death: chloroquine-resistant P. falciparum .
These brief scenarios give a sense of the diverse ways that malaria can affect people. So fundamental is this diversity with respect to impact, manifestation, and epidemiology that malaria experts themselves are not unanimous on how best to approach the disease. Malariologists recognize that malaria is essentially a local phenomenon that varies greatly from region to region and even from village to village in the same district. Consequently, a single global technology for malaria control is of little use for specific conditions, yet the task of tailoring strategies to each situation is daunting. More important, many malarious countries do not have the resources, either human or financial, to carry out even the most meager efforts to control malaria.
These scenarios also illustrate the dual nature of malaria as it affects U.S. policy. In one sense, it is a foreign aid issue; a devastating disease is currently raging out of control in vast, heavily populated areas of the world. In another sense, malaria is of domestic public health concern. The decay of global malaria control and the invasion of the parasite into previously disease-free areas, coupled with the increasing frequency of visits to such areas by American citizens, intensify the dangers of malaria for the U.S. population. Tourists, business travelers, Peace Corps volunteers, State Department employees, and military personnel are increasingly at risk, and our ability to protect and cure them is in jeopardy. What is desperately needed is a better application of existing malaria control tools and new methods of containing the disease.
In most malarious regions of the world, there is inadequate access to malaria treatment. Appropriate health facilities may not exist; those that do exist may be inaccessible to affected populations, may not be supplied with effective drugs, or may be staffed inappropriately. In many countries, the expansion of primary health care services has not proceeded according to expectations, particularly in the poorest (and most malarious) nations of the tropical world.
In some countries, antimalarial interventions are applied in broad swaths, without regard to underlying differences in the epidemiology of the disease. In other countries, there are no organized interventions at all. The malaria problem in many regions is compounded by migration, civil unrest, poorly planned exploitation of natural resources, and their frequent correlate, poverty.
During the past 15 years, much research has focused on developing vaccines for malaria. Malaria vaccines are thought to be possible in part because people who are naturally exposed to the malaria parasite acquire a partial immunity to the disease over time. In addition, immunization of animals and humans by the bites of irradiated mosquitoes infected with the malaria parasite can protect against malaria infection. Much progress has been made, but current data suggest that effective vaccines are not likely to be available for some time.
Compounding the difficulty of developing more effective malaria prevention, treatment, and control strategies is a worldwide decline in the pool of scientists and health professionals capable of conducting field research and organizing and managing malaria control programs at the country level. With the change in approach from malaria eradication to malaria control, many malaria programs “lost face,” admitting failure and losing the priority interest of their respective ministries of health. As external funding agencies lost interest in programs, they reduced their technical and financial support. As a consequence, there were fewer training opportunities, decreased contacts with international experts, and diminished prospects for improving the situation. Today, many young scientists and public health specialists, in both the developed and developing countries, prefer to seek higher-profile activities with better defined opportunities for career advancement.
It is against this backdrop of a worsening worldwide malaria situation that the Institute of Medicine was asked to convene a multidisciplinary committee to assess the current status of malaria research and control and to make recommen-
dations to the U.S. government on promising and feasible strategies to address the problem. During the 18-month study, the committee reviewed the state of the science in the major areas of malariology, identified gaps in knowledge within each of the major disciplines, and developed recommendations for future action in malaria research and control.
Organization
Chapter 2 summarizes key aspects of the individual state-of-the-science chapters, and is intended to serve as a basic introduction to the medical and scientific aspects of malaria, including its clinical signs, diagnosis, treatment, and control. Chapter 3 provides a historical overview of malaria, from roughly 3000 B.C. to the present, with special emphasis on efforts in this century to eradicate and control the disease. The state-of-the-science reviews, which start in Chapter 4 , begin with a scenario titled “Where We Want To Be in the Year 2010.” Each scenario describes where the discipline would like to be in 20 years and how, given an ideal world, the discipline would have contributed to malaria control efforts. The middle section of each chapter contains a critical review of the current status of knowledge in the particular field. The final section lays out specific directions for future research based on a clear identification of the major gaps in scientific understanding for that discipline. The committee urges those agencies that fund malaria research to consult the end of each state-of-the-science chapter for suggestions on specific research opportunities in malaria.
Sponsorship
This study was sponsored by the U.S. Agency for International Development, the U.S. Army Medical Research and Development Command, and the National Institute of Allergy and Infectious Diseases of the National Institutes of Health.
CONCLUSIONS AND RECOMMENDATIONS
A major finding of the committee is the need to increase donor and public awareness of the growing risk presented by the resurgence of malaria. Overall, funding levels are not adequate to meet the problem. The committee believes that funding in the past focused too sharply on specific technologies and particular control strategies (e.g., indiscriminate use of insecticide spraying). Future support must be balanced among the needs outlined in this report. The issue for prioritization is not whether to select specific technologies or control strategies, but to raise the priority for solv-
ing the problem of malaria. This is best done by encouraging balanced research and control strategies and developing a mechanism for periodically adjusting support for promising approaches.
This report highlights those areas which the committee believes deserve the highest priority for research or which should be considered when U.S. support is provided to malaria control programs. These observations and suggestions for future action, presented below in four sections discussing policy, research, control, and training, represent the views of a multidisciplinary group of professionals from diverse backgrounds and with a variety of perspectives on the problem.
The U.S. government is the largest single source of funds for malaria research and control activities in the world. This investment is justified by the magnitude of the malaria problem, from both a foreign aid and a public health perspective. The increasing severity of the threat of malaria to residents of endemic regions, travelers, and military personnel, and our diminishing ability to counter it, should be addressed by a more comprehensive and better integrated approach to malaria research and control. However, overall U.S. support for malaria research and control has declined over the past five years. The committee believes that the amount of funding currently directed to malaria research and control activities is inadequate to address the problem.
Over the past 10 years, the majority of U.S. funds available for malaria research have been devoted to studies on immunity and vaccine development. Although the promise of vaccines remains to be realized, the committee believes that the potential benefits are enormous. At the same time, the relative paucity of funds available for research has prevented or slowed progress in other areas. Our incomplete knowledge about the basic biology of malaria parasites, how they interact with their mosquito and human hosts, and how human biology and behavior affect malaria transmission and control remains a serious impediment to the development and implementation of malaria control strategies. The committee believes that this situation must be addressed without reducing commitment to current research initiatives. The committee further believes that such research will pay long-term dividends in the better application of existing tools and the development of new drugs, vaccines, and methods for vector control.
The committee recommends that increased funds be made available so that U.S. research on malaria can be broadened according to the priorities addressed in this report, including laboratory and field research on the biology of malaria parasites, their mosquito vectors, and their interaction with humans.
The committee believes that the maximum return on investment of funds devoted to malaria research and control can be achieved only by rigorous review of project proposals. The committee further believes that the highest-quality review is essential to ensure that funding agencies spend their money wisely. The committee believes that all U.S.-supported malaria field activities, both research and control, should be of the highest scientific quality and relevance to the goals of malaria control.
The committee recommends decisions on funding of malaria research be based on scientific merit as determined by rigorous peer review, consistent with the guidelines of the National Institutes of Health or the United Nations Development Program/World Bank/ World Health Organization Special Programme for Research and Training in Tropical Diseases, and that all U.S.-supported malaria field projects be subject to similar rigorous review to ensure that projects are epidemiologically and scientifically sound.
Commitment and Sustainability
For malaria control, short-term interventions can be expected to produce only short-term results. The committee believes that short-term interventions are justified only for emergency situations. Longer-term interventions should be undertaken only when there is a national commitment to support sustained malaria surveillance and control.
The committee recommends that malaria control programs receive sustained international and local support, oriented toward the development of human resources, the improvement of management skills, the provision of supplies, and the integration of an operational research capability in support of an epidemiologically sound approach to malaria control.
Surveillance
During the major effort to eradicate malaria from many parts of the world that began in the late 1950s and ended in 1969, it was important to establish mechanisms to detect all malaria infections. As a result, systems were established in many countries to collect blood samples for later microscopic examination for the presence of parasites. Each year, the results from more than 140 million slides are reported to the World Health Organization, of which roughly 3 to 5 percent are positive for malaria. This approach seeks to answer the question posed 30 years ago: How many people are infected with the malaria parasite? It does not answer today's questions: Who is sick? Where? Why? The committee concludes that the mass collection of blood slides requires considerable resources, poses seri-
ous biosafety hazards, deflects attention from the treatment of ill individuals, and has little practical relevance for malaria control efforts today.
Instead of the mass collection of slides, the committee believes that the most effective surveillance networks are those that concurrently measure disease in human populations, antimalarial drug use, patterns of drug resistance, and the intensity of malaria transmission by vector populations. The committee believes that malaria surveillance practices have not received adequate recognition as an epidemiologic tool for designing, implementing, and evaluating malaria control programs.
The committee recommends that countries be given support to orient malaria surveillance away from the mass collection and screening of blood slides toward the collection and analysis of epidemiologically relevant information that can be used to monitor the current situation on an ongoing basis, to identify high-risk groups, and to detect potential epidemics early in their course.
Inter-Sectoral Cooperation
The committee believes that insufficient attention has been paid to the impact that activities in non-health-related sectors, such as construction, industry, irrigation, and agriculture, have on malaria transmission. Conversely, there are few assessments of the impact of malaria control projects on other public health initiatives, the environment, and the socioeconomic status of affected populations. Malaria transmission frequently occurs in areas where private and multinational businesses and corporations (e.g., hotel chains, mining operations, and industrial plants) have strong economic interests. Unfortunately and irresponsibly, some local and multinational businesses contribute few if any resources to malaria control in areas in which they operate.
The committee recommends greater cooperation and consultation between health and nonhealth sectors in the planning and implementation of major development projects and malaria activities. It also recommends that all proposed malaria control programs be analyzed for their potential impact on other public health programs, the environment, and social and economic welfare, and that local and multinational businesses be recruited by malaria control organizations to contribute substantially to local malaria control efforts.
New Tools for Malaria Control
The committee believes that, as a policy directive, it is important to support research activities to develop new tools for malaria control. The
greatest momentum for the development of new tools exists in vaccine and drug development, and the committee believes it essential that this momentum be maintained. The committee recognizes that commendable progress has been made in defining the characteristics of antigens and delivery systems needed for effective vaccines, but that the candidates so far tested fall short of the goal. Much has been learned which supports the hope that useful vaccines can be developed. To diminish activity in vaccine development at this stage would deal a severe blow to one of our best chances for a technological breakthrough in malaria control.
The committee recommends that vaccine development continue to be a priority of U.S.-funded malaria research.
Only a handful of drugs are available to prevent or treat malaria, and the spread of drug-resistant strains of the malaria parasite threatens to reduce further the limited pool of effective drugs. The committee recognizes that there is little economic incentive for U.S. pharmaceutical companies to undertake antimalarial drug discovery activities. The committee is concerned that U.S. government support of these activities, based almost entirely at the Walter Reed Army Institute of Research (WRAIR), has decreased and is threatened with further funding cuts. The committee concludes that the WRAIR program in antimalarial drug discovery, which is the largest and most successful in the world, is crucial to international efforts to develop new drugs for malaria. The benefits of this program in terms of worldwide prevention and treatment of malaria have been incalculable.
The committee strongly recommends that drug discovery and development activities at WRAIR receive increased and sustained support.
The next recommendation on policy directions reflects the committee 's concern about the lack of involvement in malaria research by the private sector. The committee believes that the production of candidate malaria vaccines and antimalarial drugs for clinical trials has been hampered by a lack of industry involvement. Greater cooperation and a clarification of the contractual relationships between the public and private sectors would greatly enhance the development of drugs and vaccines.
The committee recommends that mechanisms be established to promote the involvement of pharmaceutical and biotechnology firms in the development of malaria vaccines, antimalarial drugs, and new tools for vector control.
Coordination and Integration
The committee is concerned that there is inadequate joint planning and coordination among U.S.-based agencies that support malaria research and
control activities. Four government agencies and many nongovernmental organizations in the United States are actively involved in malaria-related activities. There are also numerous overseas organizations, governmental and nongovernmental, that actively support such activities worldwide.
The complexity and variability of malaria, the actual and potential scientific advances in several areas of malariology, and most important the worsening worldwide situation argue strongly for an ongoing mechanism to assess and influence current and future U.S. efforts in malaria research and control.
The committee strongly recommends the establishment of a national advisory body on malaria.
In addition to fulfilling a much needed coordinating function among U.S.-based agencies and between the U.S. and international efforts, the national advisory body could monitor the status of U.S. involvement in malaria research and control, assess the relevant application of knowledge, identify areas requiring further research, make recommendations to the major funding agencies, and provide a resource for legislators and others interested in scientific policy related to malaria. The national advisory body could convene specific task-oriented scientific working groups to review research and control activities and to make recommendations, when appropriate, for changes in priorities and new initiatives.
The committee believes that the national advisory body should be part of, and appointed by, a neutral and nationally respected scientific body and that it should actively encourage the participation of governmental and nongovernmental organizations, industry, and university scientists in advising on the direction of U.S. involvement in malaria research and control.
The increasing magnitude of the malaria problem during the past decade and the unpredictability of changes in human, parasite, and vector determinants of transmission and disease point strongly to the need for such a national advisory body, which can be responsive to rapidly changing problems, and advances in scientific research, relating to global efforts to control malaria.
Malaria Research Priorities
Malaria control is in crisis in many areas of the world. People are contracting and dying of severe malaria in unprecedented numbers. To address these problems, the committee strongly encourages a balanced research agenda. Two basic areas of research require high priority. Research that will lead to improved delivery of existing interventions for malaria, and the development of new tools for the control of malaria.
Research in Support of Available Control Measures
Risk Factors for Severe Malaria People who develop severe and complicated malaria lack adequate immunity, and many die from the disease. Groups at greatest risk include young children and pregnant women in malaria endemic regions; nonimmune migrants, laborers, and visitors to endemic regions; and residents of regions where malaria has been recently reintroduced. For reasons that are largely unknown, not all individuals within these groups appear to be at equal risk for severe disease. The committee believes that the determinants of severe disease, including risk factors associated with a population, the individual (biologic, immunologic, socioeconomic, and behavioral), the parasite, or exposure to mosquitoes, are likely to vary considerably in different areas.
The committee recommends that epidemiologic studies on the risk factors for severe and complicated malaria be supported.
Pathogenesis of Severe and Complicated Malaria Even with optimal care, 20 to 30 percent of children and adults with the most severe form of malaria—primarily cerebral malaria—die. The committee believes that a better understanding of the disease process will lead to improvements in preventing and treating severe forms of malaria. The committee further believes that determining the indications for treatment of severe malarial anemia is of special urgency given the risk of transmitting the AIDS virus through blood transfusions, the only currently available treatment for malarial anemia. Physicians need to know when it is appropriate to transfuse malaria patients.
The committee recommends greater support for research on the pathogenesis of severe and complicated malaria, on the mechanisms of malarial anemia, and on the development of specific criteria for blood transfusions in malaria.
Social Science Research The impact of drugs to control disease or programs to reduce human-mosquito contact is mediated by local practices and beliefs about malaria and its treatment. Most people in malaria-endemic countries seek initial treatment for malaria outside of the formal health sector. Programs that attempt to influence this behavior must understand that current practices satisfy, at some level, local concerns regarding such matters as access to and effectiveness of therapy, and cost. These concerns may lead to practices at odds with current medical practice. Further, many malaria control programs have not considered the social, cultural, and behavioral dimensions of malaria, thereby limiting the effectiveness of measures undertaken. The committee recognizes that control programs often fail to incorporate household or community concerns and resources
into program design. In most countries, little is known about how the demand for and utilization of health services is influenced by such things as user fees, location of health clinics, and the existence and quality of referral services. The committee concludes that modern social science techniques have not been effectively applied to the design, implementation, and evaluation of malaria control programs.
The committee recommends that research be conducted on local perceptions of malaria as an illness, health-seeking behaviors (including the demand for health care services), and behaviors that affect malaria transmission, and that the results of this research be included in community-based malaria control interventions that promote the involvement of communities and their organizations in control efforts.
Innovative Approaches to Malaria Control Malaria control programs will require new ideas and approaches, and new malaria control strategies need to be developed and tested. There is also a need for consistent support of innovative combinations of control technologies and for the transfer of new technologies from the laboratory to the clinic and field for expeditious evaluation. Successful technology transfer requires the exchange of scientific research, but more importantly, must be prefaced by an improved understanding of the optimal means to deliver the technology to the people in need (see Chapter 11 ).
The committee recommends that donor agencies provide support for research on new or improved control strategies and into how new tools and technologies can be better implemented and integrated into on-going control efforts.
Development of New Tools
Antimalarial Immunity and Vaccine Development Many people are able to mount an effective immune response that can significantly mitigate symptoms of malaria and prevent death. The committee believes that the development of effective malaria vaccines is feasible, and that the potential benefits of such vaccines are enormous. Several different types of malaria vaccines need to be developed: vaccines to prevent infection (of particular use for tourists and other nonimmune visitors to endemic countries), prevent the progression of infection to disease (for partially immune residents living in endemic areas and for nonimmune visitors), and interrupt transmission of parasites by vector populations (to reduce the risk of new infections in humans). The committee believes that each of these directions should be pursued.
The committee recommends sustained support for research to identify mechanisms and targets of protective immunity and to exploit the
use of novel scientific technologies to construct vaccines that induce immunity against all relevant stages of the parasite life cycle.
Drug Discovery and Development Few drugs are available to prevent or treat malaria, and the spread of drug-resistant strains of malaria parasites is steadily reducing the limited pool of effective chemotherapeutic agents. The committee believes that an inadequate understanding of parasite biochemistry and biology impedes the process of drug discovery and slows studies on the mechanisms of drug resistance.
The committee recommends increased emphasis on screening compounds to identify new classes of potential antimalarial drugs, identifying and characterizing vulnerable targets within the parasite, understanding the mechanisms of drug resistance, and identifying and developing agents that can restore the therapeutic efficacy of currently available drugs.
Vector Control Malaria is transmitted to humans by the bites of infective mosquitoes. The objective of vector control is to reduce the contact between humans and infected mosquitoes. The committee believes that developments are needed in the areas of personal protection, environmental management, pesticide use and application, and biologic control, as well as in the largely unexplored areas of immunologic and genetic approaches for decreasing parasite transmission by vectors.
The committee recommends increased support for research on vector control that focuses on the development and field testing of methods for interrupting parasite transmission by vectors.
Malaria Control
Malaria is a complex disease that, even under the most optimistic scenario, will continue to be a major health threat for decades. The extent to which malaria affects human health depends on a large number of epidemiologic and ecologic factors. Depending on the particular combination of these and other variables, malaria may have different effects on neighboring villages and people living in a single village. All malaria control programs need to be designed with a view toward effectiveness and sustainability, taking into account the local perceptions, the availability of human and financial resources, and the multiple needs of the communities at risk. If community support for health sector initiatives is to be guaranteed, the public needs to know much more about malaria, its risks for epidemics and severe disease, and difficulties in control.
Unfortunately, there is no “magic bullet” solution to the deteriorating worldwide malaria situation, and no single malaria control strategy will be applicable in all regions or epidemiologic situations. Given the limited available financial and human resources and a dwindling pool of effective
antimalarial tools, the committee suggests that donor agencies support four priority areas for malaria control in endemic countries.
The committee believes that the first and most basic priority in malaria control is to prevent infected individuals from becoming severely ill and dying. Reducing the incidence of severe morbidity and malaria-related mortality requires a two-pronged approach. First, diagnostic, treatment, and referral capabilities, including the provision of microscopes, training of technicians and other health providers, and drug supply, must be enhanced. Second, the committee believes that many malaria-related deaths could be averted if individuals and caretakers of young children knew when and how to seek appropriate treatment and if drug vendors, pharmacists, physicians, nurses, and other health care providers were provided with up-to-date and locally appropriate treatment and referral guidelines. The development and implementation of an efficient information system that provides rapid feedback to the originating clinic and area is key to monitoring the situation and preventing epidemics.
The committee believes that the second priority should be to promote personal protection measures (e.g., bednets, screens, and mosquito coils) to reduce or eliminate human-mosquito contact and thus to reduce the risk of infection for individuals living in endemic areas. At the present time, insecticide-treated bednets appear to be the most promising personal protection method.
In many environments, in addition to the treatment of individuals and use of personal protection measures, community-wide vector control is feasible. In such situations, the committee believes that the third priority should be low-cost vector control measures designed to reduce the prevalence of infective mosquitoes in the environment, thus reducing the transmission of malaria to populations. These measures include source reduction (e.g., draining or filling in small bodies of water where mosquito larvae develop) or the application of low-cost larval control measures. In certain environments, the use of insecticide-impregnated bednets by all or most members of a community may also reduce malaria transmission, but this approach to community-based malaria control remains experimental.
The committee believes that the fourth priority for malaria control should be higher cost vector control measures such as large-scale source reduction or widespread spraying of residual insecticides. In certain epidemiologic situations, the use of insecticides for adult mosquito control is appropriate and represents the method of choice for decreasing malaria transmission and preventing epidemics (see Chapter 7 and Chapter 10 ).
The committee recommends that support of malaria control programs include resources to improve local capacities to conduct prompt diagnosis, including both training and equipment, and to ensure the availability of antimalarial drugs.
The committee recommends that resources be allocated to develop and disseminate malaria treatment guidelines for physicians, drug vendors, pharmacists, village health workers, and other health care personnel in endemic and non-endemic countries. The guidelines should be based, where appropriate, on the results of local operational research and should include information on the management of severe and complicated disease. The guidelines should be consistent and compatible among international agencies involved in the control of malaria.
The committee recommends that support for malaria control initiatives include funds to develop and implement locally relevant communication programs that provide information about how to prevent and treat malaria appropriately (including when and how to seek treatment) and that foster a dialogue about prevention and control.
Organization of Malaria Control
One of the major criticisms of malaria control programs during the past 10 to 15 years has been that funds have been spent inappropriately without an integrated plan and without formal evaluation of the efficacy of control measures instituted. In many instances, this has led to diminished efforts to control malaria.
The committee strongly encourages renewed commitment by donor agencies to support national control programs in malaria-endemic countries.
The committee recommends that U.S. donor agencies develop, with the advice of the national advisory body, a core of expertise (either in-house or through an external advisory group) to plan assistance to malaria control activities in endemic countries.
The committee believes that the development, implementation, and evaluation of such programs must follow a rigorous set of guidelines. These guidelines should include the following steps:
Identification of the problem
Determine the extent and variety of malaria. The paradigm approach described in Chapter 10 should facilitate this step.
Analyze current efforts to solve malaria problems.
Identify and characterize available in-country resources and capabilities.
Development of a plan
Design and prioritize interventions based on the epidemiologic situation and the available resources.
Design a training program for decision makers, managers, and technical staff to support and sustain the interventions.
Define specific indicators of the success or failure of the interventions at specific time points.
Develop a specific plan for reporting on the outcomes of interventions.
Develop a process for adjusting the program in response to successes and/or failures of interventions.
Review of the comprehensive plan by a donor agency review board
Modification of the plan based on comments of the review board
Implementation of the program
Yearly report and analysis of outcome variables
To guide the implementation of the activities outlined above, the committee has provided specific advice on several components, including an approach to evaluating malaria problems and designing control strategies (the paradigm approach), program management, monitoring and evaluation, and operational research.
Paradigm Approach
Given the complex and variable nature of malaria, the committee believes that the epidemiologic paradigms (see Chapter 10 ), developed in conjunction with this study, may form the basis of a logical and reasoned approach for defining the malaria problems and improving the design and management of malaria control programs.
The committee recommends that the paradigm approach be field tested to determine its use in helping policymakers and malaria program managers design and implement epidemiologically appropriate and cost-effective control initiatives.
The committee recognizes that various factors, including the local ecology, the dynamics of mosquito transmission of malaria parasites, genetically determined resistance to malaria infection, and patterns of drug use, affect patterns of malaria endemicity in human populations and need to be considered when malaria control strategies are developed. In most endemic countries, efforts to understand malaria transmission through field studies of vector populations are either nonexistent or so limited in scope that they have minimal impact on subsequent malaria control efforts. The committee recognizes that current approaches to malaria control are clearly inadequate. The committee believes, however, that malaria control strategies are sometimes applied inappropriately, with little regard to the underlying differences in the epidemiology of the disease.
The committee recommends that support for malaria control programs include funds to permit a reassessment and optimization of antimalarial tools based on relevant analyses of local epidemiologic, parasitologic, entomologic, socioeconomic, and behavioral determinants of malaria and the costs of malaria control.
Poor management has contributed to the failure of many malaria control programs. Among the reasons are a chronic shortage of trained managers who can think innovatively about health care delivery and who can plan, implement, supervise, and evaluate malaria control programs. Lack of incentives, the absence of career advancement options, and designation of responsibility without authority often hinder the effectiveness of the small cadre of professional managers that does exist. The committee recognizes that management technology is a valuable resource that has yet to be effectively introduced into the planning, implementation, and evaluation of most malaria control programs.
The committee recommends that funding agencies utilize management experts to develop a comprehensive series of recommendations and guidelines as to how basic management skills and technology can be introduced into the planning, implementation, and evaluation of malaria control programs.
The committee recommends that U.S. funding of each malaria control program include support for a senior manager who has responsibility for planning and coordinating malaria control activities. Where such an individual does not exist, a priority of the control effort should be to identify and support a qualified candidate. The manager should be supported actively by a multidisciplinary core group with expertise in epidemiology, entomology, the social sciences, clinical medicine, environmental issues, and vector control operations.
Monitoring and Evaluation
Monitoring and evaluation are essential components of any control program. For malaria control, it is not acceptable to continue pursuing a specific control strategy without clear evidence that it is effective and reaching established objectives.
The committee recommends that support for malaria control programs include funds to evaluate the impact of control efforts on the magnitude of the problem and that each program be modified as necessary on the basis of periodic assessments of its costs and effectiveness.
Problem Solving (Operational Research) and Evaluation
At the outset of any malaria prevention or control initiative and during the course of implementation, gaps in knowledge will be identified and problems will arise. These matters should be addressed through clearly defined, short-term, focused studies. Perhaps the most difficult aspects of operational research are to identify the relevant problem, formulate the appropriate question, and design a study to answer that question.
The committee recommends that a problem-solving (operational research) component be built into all existing and future U.S.-funded malaria control initiatives and that support be given to enhance the capacity to perform such research. This effort will include consistent support in the design of focused projects that can provide applicable results, analysis of data, and dissemination of conclusions.
The committee concludes that there is a need for additional scientists actively involved in malaria-related research in the United States and abroad. To meet this need, both short- and long-term training at the doctoral and postdoctoral levels must be provided. This training will be of little value unless there is adequate long-term research funding to support the career development of professionals in the field of malaria.
The committee recommends support for research training in malaria.
Whereas the curricula for advanced degree training in basic science research and epidemiology are fairly well defined, two areas require attention, especially in the developing world: social sciences and health management and training.
The committee recommends that support be given for the development of advanced-degree curricula in the social sciences, and in health management and training, for use in universities in developing and developed countries.
The availability of well-trained managers, decision makers, and technical staff is critical to the implementation of any malaria prevention and control program. The development of such key personnel requires a long term combination of formal training, focused short courses, and a gradual progression of expertise.
The committee recommends support for training in management, epidemiology, entomology, social sciences, and vector control. Such training for malaria control may be accomplished through U.S.-funded grant programs for long-term cooperative relationships
between institutions in developed and developing countries; through the encouragement of both formal and informal linkages among malaria-endemic countries; through the use of existing training courses; and through the development of specific training courses.
The committee recommends further that malaria endemic countries be supported in the development of personnel programs that provide long-term career tracks for managers, decision makers, and technical staff, and that offer professional fulfillment, security, and competitive financial compensation.
Malaria is making a dramatic comeback in the world. The disease is the foremost health challenge in Africa south of the Sahara, and people traveling to malarious areas are at increased risk of malaria-related sickness and death.
This book examines the prospects for bringing malaria under control, with specific recommendations for U.S. policy, directions for research and program funding, and appropriate roles for federal and international agencies and the medical and public health communities.
The volume reports on the current status of malaria research, prevention, and control efforts worldwide. The authors present study results and commentary on the:
- Nature, clinical manifestations, diagnosis, and epidemiology of malaria.
- Biology of the malaria parasite and its vector.
- Prospects for developing malaria vaccines and improved treatments.
- Economic, social, and behavioral factors in malaria control.
READ FREE ONLINE
Welcome to OpenBook!
You're looking at OpenBook, NAP.edu's online reading room since 1999. Based on feedback from you, our users, we've made some improvements that make it easier than ever to read thousands of publications on our website.
Do you want to take a quick tour of the OpenBook's features?
Show this book's table of contents , where you can jump to any chapter by name.
...or use these buttons to go back to the previous chapter or skip to the next one.
Jump up to the previous page or down to the next one. Also, you can type in a page number and press Enter to go directly to that page in the book.
Switch between the Original Pages , where you can read the report as it appeared in print, and Text Pages for the web version, where you can highlight and search the text.
To search the entire text of this book, type in your search term here and press Enter .
Share a link to this book page on your preferred social network or via email.
View our suggested citation for this chapter.
Ready to take your reading offline? Click here to buy this book in print or download it as a free PDF, if available.
Get Email Updates
Do you enjoy reading reports from the Academies online for free ? Sign up for email notifications and we'll let you know about new publications in your areas of interest when they're released.
- Meeting report
- Open access
- Published: 27 May 2022
Reflections on the 2021 World Malaria Report and the future of malaria control
- April Monroe 1 , 7 ,
- Nana Aba Williams 2 , 3 ,
- Sheila Ogoma 4 ,
- Corine Karema 5 , 6 &
- Fredros Okumu 7
Malaria Journal volume 21 , Article number: 154 ( 2022 ) Cite this article
11k Accesses
44 Citations
68 Altmetric
Metrics details
The World Malaria Report, released in December 2021, reflects the unique challenges currently facing the global malaria community. The report showed the devastating toll of malaria, with an estimated 627,000 people losing their lives to the disease in 2020. The improved methodological approach used for calculating cause of death for young children revealed a systematic underestimation of disease burden over the past two decades; and that Africa has an even greater malaria crisis than previously known. While countries were able to prevent the worst-case scenarios, the disruptions due to the COVID-19 pandemic revealed how weak health systems and inadequate financing can limit the capacity of the continent to address the malaria challenge. African countries also face a convergence of biological threats that could redefine malaria control, notably widespread pyrethroid resistance and emerging resistance to artemisinin. Despite these challenges, there is cause for optimism in lessons learned from the COVID-19 pandemic, recent acceleration of cutting edge research and development, and new partnerships that encourage leadership from and ownership by affected countries. This article presents key insights from the 2021 World Malaria Report and reflections on the future trajectories: it was informed by an in-depth discussion with leading malaria experts from the World Health Organization (WHO), the Bill & Melinda Gates Foundation, and the U.S. President’s Malaria Initiative (PMI). The discussion took place during the 34th edition of the Ifakara Master Classes, held virtually on December 15th, 2021.
On December 15th, 2021, the 34th edition of the Ifakara Master Classes featured an in-depth discussion on the 2021 World Malaria Report (WMR), released a week earlier [ 1 ]. The discussion unpacked WMR findings and their implications for the future of malaria control. Guest experts included Dr. Pedro Alonso, Director of the Global Malaria Programme (GMP) at the World Health Organization (WHO), Dr. Abdisalan Noor, WHO Head of Strategic Information for Response Unit, Dr. Jennifer Gardy, Deputy Director, Surveillance, Data, and Epidemiology at the Bill & Melinda Gates Foundation, and Dr. Richard Steketee, Deputy Global Malaria Coordinator for the U.S. President’s Malaria Initiative (PMI).
The discussion, which lasted 2 h and 45 min in total, was organized and facilitated by MasterClass hosts Drs. Fredros Okumu (Director of Science, Ifakara Health Institute, Tanzania) and Sheila Ogoma (Technical Director, Clinton Health Access Initiative), and guest hosts, Drs. Corine Karema (Private Consultant and former Director of National Malaria Control Programme, Rwanda) and Nana Aba Williams (Coordinator, MESA Alliance, ISGlobal, Spain). The session began with a brief overview of the 2021 WMR by Dr. Noor, followed by a series of open-ended technical questions posed by the facilitators to the panel of experts about specific aspects of the WMR. The discussion was hosted on Zoom with 320 live participants from the global malaria community, and was live-streamed on YouTube.
A consolidated account of insights and lessons learned from the discussion is presented here. Findings are organized around topics identified a priori by the Master Class facilitators and key themes that emerged through the discussion.
The importance of numbers
The World Malaria Report, released December 2021, reflects the unique challenges facing the global malaria community. The report lays bare the devastating toll of malaria, with an estimated 627,000 people losing their lives to the disease in 2020. The numbers in the report tell two different stories for countries nearing elimination and countries experiencing high burden.
“A growing number of countries with low burden are moving steadily toward elimination, while countries with the highest burden are struggling.” –Dr. Noor
Eleven countries now experience 70% of the world’s malaria burden while 47 now report fewer than 10,000 cases per year. Even before the COVID-19 pandemic, gains against malaria were leveling off, leading to the role out of the High burden, High impact response in 2018 [ 2 ].
Methodological changes
A new statistical method is being used by the WHO, which provides more precise cause-of-death estimates for young children for all diseases, including malaria. In the revised approach, the proportion of childhood deaths attributable to malaria was 7.8%, up from previous estimates of 4.8% [ 3 , 4 ]. The revised approach revealed that there had been a higher number of estimated deaths between 2000 and 2020 than previously recognized and a systematic underestimation across the time series. The revisions also suggest that a higher number of malaria cases (totaling 1.7 billion) and deaths (10.6 million) had been averted in the same period.
The WMR has gotten clearer, and the quality improved consistently since it was first released. However, for most countries, the WHO still relies on modelled estimates derived from verbal autopsies to calculate all-cause mortality and the cause of death fraction for children under-5 to quantify malaria deaths in this age-group before applying a second adjustment to quantify deaths in older children and adults. There is a strong case for improving surveillance as an intervention and investing more heavily in information systems as recommended in the WHO Global Technical Strategy (GTS) 2016–2030 [ 5 ]. These malaria metrics, whether estimates or not, can be powerful advocacy tools and are, therefore, integral for creating compelling narratives of changes over time.
Impact of COVID-19
In addition to increases due to the methodological changes, the COVID-19 pandemic posed significant challenges, and was associated with ~ 47,000 of the ~ 69,000 extra deaths reported in 2020 relative to 2019, [ 1 ]. This includes increases in cases due to disruptions associated with delays in ITN distribution and disruptions in both diagnosis and treatment. Malaria deaths increased by 12% to an estimated 627,000 in 2020, compared to 2019 figures, with more than two-thirds of the additional 69,000 deaths attributable to COVID19-related service disruptions. While the figures are worrying, countries and partners have done well to prevent the worst-case scenarios earlier projected by the WHO and partners [ 6 , 7 , 8 ]; these models had predicted increases in malaria cases and deaths in Africa of as much as two orders of magnitude.
Threats to malaria control in Africa
A range of challenges from biological threats, to preventing severe disease and death in the most remote areas, to fragile and insufficient malaria funding must be addressed to sustain progress.
“ The situation remains precarious, particularly in sub-Saharan Africa where burden remains unacceptably high and a convergence of threats pose added challenges to disease control efforts…Without immediate accelerated action, key 2030 targets of the WHO Global Technical Strategy [ 5 ] for malaria will be missed, and additional ground may be lost.” –Dr. Noor
- Biological threats
While the epidemiology of malaria in Africa is already more challenging and precarious than elsewhere, the situation is compounded by multiple biological and civil threats. Over 122 million people in 21 malaria-endemic countries needed assistance due to health and humanitarian emergencies in 2020–2021 including Ebola outbreaks, armed conflicts, and flooding. Key biological threats in sub-Saharan Africa include anti-malarial drug resistance in the eastern Africa region [ 9 , 10 , 11 ], threats to diagnostics posed by parasite pfhrp2/3 gene deletions (which can cause false negative diagnostic test results) [ 12 , 13 ], resistance of malaria vector mosquitoes to public health insecticides [ 14 , 15 ], and the invasive vector species, Anopheles stephensi in the Horn of Africa [ 16 , 17 , 18 ]. All these factors threaten to undermine malaria control efforts in ways that are not sufficiently understood.
The WHO is tracking biological threats using the WHO threats map [ 19 ]. For pfhrp2/3 gene deletions, there are already new tests, albeit more expensive, which are prequalified by the WHO that can detect these parasites [ 20 ]. Increased investments to improve surveillance of gene deletions is needed and investments in new diagnostics is essential and a cause for optimism. Insecticide resistance remains a significant challenge to be addressed decisively—PBO nets are now recommended, and other new generation nets are being evaluated [ 21 ]. The WHO recognizes A. stephensi as an efficient malaria vector in urban settings [ 22 ], and affected countries and their neighbours should urgently enhance surveillance and deploy novel tools. Given these threats, malaria stakeholders should be open to examining other potentially-transformative approaches such as genetically modified mosquitoes currently in early-stage development [ 23 , 24 ].
Of particular concern is emerging signs of resistance to artemisinin, which is the backbone of current malaria treatment efforts in Africa [ 9 , 10 , 11 ]. Now confirmed in Uganda [ 9 ] and Rwanda [ 10 , 11 ], artemisinin resistance, more accurately described as delayed parasite clearance, is emerging de novo in Africa and does not appear to be linked to the resistance in malaria parasites in south-east Asia, where this problem was first described [ 25 ]. Setting up effective surveillance systems is, therefore, critical to closely track this threat in the region.
Severe malaria and the last mile
Combatting severe malaria is paramount for averting malaria deaths and depends on systems that support prompt treatment, referral for severe disease, and a full course of treatment to clear infection. However, the most severe malaria cases and deaths are often concentrated in areas where health systems are weakest, where prevention practices are most inadequate, and care workers least trained. Effective community-based approaches, particularly training and appropriately compensating community health workers will be key to reaching the unreached and preventing severe disease.
“…This is a Catch 22… if we try to build our health systems to reach the people furthest out, and at the greatest risk, using our least trained, least supplied workers, the system is then going to have to deal with severe malaria because we weren’t able to prevent it in the first place...the question is, how do we take the community outreach, and community health workers on the periphery, and make sure they’re sufficient in scale, have the right skills, and that they are adequately supervised and supplied?” –Dr. Steketee
Funding gap
A consistent feature of global malaria programmes is that less than half of the necessary annual budget is actually available. A total of $3.3 billion was invested in 2020, compared to target of $6.8 billion. Moreover, to reach global targets, investments will need to increase by more than three times by 2030 to 10.3 billion per year. The current system relies on just a small number of major funders and budget needs are unlikely to be met even if these few sources increase their contributions. Further, the relative investment of countries has not increased despite economic growth.
“When you think about what’s stalled, population growth has not stalled, and that will continue, what’s stalled is the money. We’ve been working on efficiencies but there are limits to what we can achieve with efficiency alone.” –Dr. Steketee
The future of malaria control
The malaria situation cannot be effectively tackled using current practices, highlighting the need for a more transformational approach, tailored to different epidemiological contexts. A drastic change in mindset is needed around the disease and its complexities.
“It has not sunk in that we need to do something drastically different. It is a mindset problem, we need to show greater flexibility, and understand we are facing a very complex problem…malaria is a problem to be solved, not simply a task to be performed.” –Dr. Alonso
Lessons learned from the COVID-19 pandemic
There are important opportunities to learn from the COVID-19 pandemic. The pandemic brought the global malaria community together in a way not previously seen, to ensure a buffer against service delivery disruptions.
“It was really heartening to see that when there’s an emergency, we can work effectively across stakeholders to mount an effective response. COVID19 responses have also demonstrated to Ministries of Health that data matters – high-quality real-time data matters.” –Dr. Gardy
The pandemic has also shown that molecular data can provide important information on current and evolving trends over time, and that mathematical models can be valuable for exploring different intervention scenarios, an approach that is now also being utilized in the WHO-backed High burden, High impact response [ 2 ]. Perhaps most promising has been lessons learned from the development of the COVID-19 vaccine.
“...We’ve seen that things like a massive investment in de-risking multiple aspects of the vaccine production pipeline meant that you could very quickly get new products authorized, under Emergency Use Authorizations, and then eventually under full approval for use. We also saw the culmination of decades of work on mRNA vaccines…it’s working better than what we could have imagined. To hear that there’s now an mRNA pipeline for malaria vaccines is very exciting.” –Dr. Gardy
Innovative financing mechanisms will be needed moving forward to ensure sufficient and sustained funding. Resource mobilization seen during the COVID-19 pandemic shows when can be done when a disease is viewed as a global threat.
“COVID-19 may provide an opportunity – when countries in the global north have felt threatened there’s no limit to the money they spend – building on this momentum is a great opportunity to put the health agenda up front. Strengthening health systems is a key issue in the fight against malaria, it may not be considered malaria money, but is key to getting the commodities out.” –Dr. Alonso
RTS,S malaria vaccine
In 2021, the RTS,S malaria vaccine became the first to be approved for widespread use; and the only vaccine currently available for any human malaria parasites. The vaccine is now recommended for children living in areas with moderate to high Plasmodium falciparum transmission. In addition to the modest efficacy demonstrated in earlier clinical trials [ 26 , 27 ] and results of a consensus modelling programme [ 28 ], data from a WHO-backed pilot study in three countries, Kenya, Ghana and Malawi, suggest that the vaccine is feasible to deliver, safe and has a significant public health impact [ 29 ]. When provided in the context of both the expanded programme of childhood immunizations and other malaria control efforts, the vaccine increases access to prevention for vulnerable children—for instance reaching two thirds of children not protected by insecticide-treated nets (ITNs)—and is cost effective in areas with moderate to high transmission. The vaccine programme has already reached more than 900,000 children in three countries and generated among the most robust evidence for a malaria control tool ever.
It will be critical to think comprehensively about malaria control, including the vaccine, to ensure context-appropriate packages of interventions.
“…Putting one tool against another is really unhelpful, it’s bad public health…We have an armamentarium, we have a set of tools, and we need to look at what’s best in a particular circumstance….” –Dr. Alonso
During the evaluation of RTS,S there was a strong partnership between African scientists, the WHO, and several other players working jointly. For example, the Phase III trials were done in 11 different sites across nine African countries [ 26 , 27 ], and the mathematical modelling done to support final decision-making had been conducted jointly by four different research groups [ 28 ]. There is an important opportunity to leverage benefits of such united approaches to improve outcomes for other technologies and malaria control programmes.
“RTS,S forces the malaria community to work with other departments of the ministry of health that are the custodians of the delivery platforms, such as EPI. Therefore, an added benefit of RTS,S is that it will force the malaria community to come out from a siloed space.” –Dr. Alonso
Working across disease portfolios can also maximize efficiencies in health systems.
“The more we can figure out how to work together on delivery platforms, the more we can see benefits across the board and use the limited (funding) envelope more effectively.” –Dr. Gardy
Toward a unified vision and country-led decision-making
Finally, the future of malaria control will require moving toward country-led, unified visions and funding strategies. This includes ensuring evidence-based decisions and centering affected countries in those decisions.
“If a country has the data to show an area would benefit from a fifth round of seasonal malaria chemoprevention, who is anyone on this planet to tell them no? We need to break those attitudes, the lack of empowerment to countries, that lack of evidence-based decision making – only then will we be able to make progress.” –Dr. Alonso
There must also be a more coordinated response from different partners working within countries and a united strategy. This includes movement toward a single national strategic plan, that is costed properly and against which the investments from inside and outside of the country are aligned to achieve the agreed programme goals.
“Let’s get everyone at the table with one single plan, costed, that we all help develop and that we can all invest in. This is truly a partnership.” –Dr. Steketee
The global malaria community is at an inflection point; progress has levelled off and multiple threats confront countries already hardest hit by the disease. A shift in mindset is urgently needed with truly innovative and collaborative approaches to malaria control. Reflecting on the 2021 WMR and its implications for the future, there is a critical opportunity to take-up lessons learned from the COVID-19 pandemic, including what is possible when the world comes together towards a common goal. Cutting edge research and development, as was seen in recent vaccine development, and improved surveillance, can pave the way to more transformational approaches. Finally, and most importantly, the future of malaria control must be led by affected countries, with unified and coordinated support from donors and partners.
Data availability
Not applicable.
WHO. World malaria report. Geneva: World Health Organization; 2021.
Google Scholar
WHO. High burden to high impact: a targeted malaria response. Geneva: World Health Organization; 2018.
Perin J, Mulick A, Yeung D, Villavicencio F, Lopez G, Strong K, et al. Global, regional, and national causes of under-5 mortality in 2000–19: an updated systematic analysis with implications for the sustainable development goals. Lancet Child Adolesc Health. 2021;6:106–15.
Article Google Scholar
WHO. Meeting report of the WHO evidence review group on malaria burden estimation methods. Geneva: World Health Organization; 2018.
WHO. Global technical strategy for malaria 2016–2030. Geneva: World Health Organization; 2015.
WHO. The potential impact of health service disruptions on the burden of malaria: a modelling analysis for countries in sub-Saharan Africa. Geneva: World Health Organization; 2020.
Sherrard-Smith E, Hogan AB, Hamlet A, Watson OJ, Whittaker C, Winskill P, et al. The potential public health consequences of COVID-19 on malaria in Africa. Nat Med. 2020;26:1411–6.
Article CAS Google Scholar
Weiss DJ, Bertozzi-Villa A, Rumisha SF, Amratia P, Arambepola R, Battle KE, et al. Indirect effects of the COVID-19 pandemic on malaria intervention coverage, morbidity, and mortality in Africa: a geospatial modelling analysis. Lancet Infect Dis. 2021;21:59–69.
Balikagala B, Fukuda N, Ikeda M, Katuro OT, Tachibana S, Yamauchi M, et al. Evidence of artemisinin-resistant malaria in Africa. N Engl J Med. 2021;385:1163–71.
Uwimana A, Legrand E, Stokes BH, Ndikumana JM, Warsame M, Umulisa N, et al. Emergence and clonal expansion of in vitro artemisinin-resistant Plasmodium falciparum kelch13 R561H mutant parasites in Rwanda. Nat Med. 2020;26:1602–8.
Uwimana A, Umulisa N, Venkatesan M, Svigel SS, Zhou Z, Munyaneza T, et al. Association of Plasmodium falciparum kelch13 R561H genotypes with delayed parasite clearance in Rwanda: an open-label, single-arm, multicentre, therapeutic efficacy study. Lancet Infect Dis. 2021;21:1120–8.
Feleke SM, Reichert EN, Mohammed H, Brhane BG, Mekete K, Mamo H, et al. Plasmodium falciparum is evolving to escape malaria rapid diagnostic tests in Ethiopia. Nat Microbiol. 2021;6:1289–99.
Alemayehu GS, Blackburn K, Lopez K, Dieng CC, Lo E, Janies D, et al. Detection of high prevalence of Plasmodium falciparum histidine-rich protein 2/3 gene deletions in Assosa zone, Ethiopia: implication for malaria diagnosis. Malar J. 2021;20:109.
Hancock PA, Hendriks CJM, Tangena J, Gibson H, Hemingway J, Coleman M, et al. Mapping trends in insecticide resistance phenotypes in African malaria vectors. PLoS Biol. 2020;18: e3000633.
Hemingway J. Resistance: a problem without an easy solution. Pestic Biochem Physiol. 2018;151:73–5.
Ahmed A, Khogali R, Elnour MB, Nakao R, Salim B. Emergence of the invasive malaria vector Anopheles stephensi in Khartoum state central Sudan. Parasit Vectors. 2021;14:511.
Sinka M, Pironon S, Massey NC, Longbottom J, Hemingway J, Moyes CL, et al. A new malaria vector in Africa: predicting the expansion range of Anopheles stephensi and identifying the urban populations at risk. Proc Natl Acad Sci USA. 2020;117:24900–8.
Takken W, Lindsay S. Increased threat of urban malaria from Anopheles stephensi mosquitoes Africa. Emerg Infect Dis. 2019;25:1431–3.
WHO. Malaria threats map: Tracking biological challenges to malaria control and elimination. Geneva, World Health Organization, 2021 [cited 2022 January 2022]; Available from: https://apps.who.int/malaria/maps/threats/ ?
WHO. Prequalified In Vitro diagnostics. Geneva, World Health Organization, 2022 [cited 2022 January]; Available from: https://extranet.who.int/pqweb/vitro-diagnostics/vitro-diagnostics-lists .
WHO. Prequalification vector control: Prequalified lists of vector control products. Geneva, World Health Organization, 2021 [cited 2021 March]; Available from: https://extranet.who.int/pqweb/vector-control-products .
WHO. Vector alert: Anopheles stephensi invasion and spread: Horn of Africa, the Republic of the Sudan and surrounding geographical areas, and Sri Lanka: information note. Geneva: World Health Organization; 2019.
Gantz VM, Jasinskiene N, Tatarenkova O, Fazekas A, Macias VM, Bier E, et al. Highly efficient Cas9-mediated gene drive for population modification of the malaria vector mosquito Anopheles stephensi . Proc Natl Acad Sci USA. 2015;112:E6736–43.
Nolan T. Control of malaria-transmitting mosquitoes using gene drives. Philos Trans R Soc B. 2021;376:20190803.
Dondorp AM, Nosten F, Yi P, Das D, Phyo AP, Tarning J, et al. Artemisinin resistance in Plasmodium falciparum malaria. N Engl J Med. 2009;361:455–67.
RTSS Clinical Trials Partnership. First results of phase 3 trial of RTS, S/AS01 malaria vaccine in African children. N Engl J Med. 2011;365:1863–75.
RTSS Clinical Trials Partnership. Efficacy and safety of the RTS, S/AS01 malaria vaccine during 18 months after vaccination: a phase 3 randomized, controlled trial in children and young infants at 11 African sites. PLoS Med. 2014;11: e1001685.
Penny MA, Verity R, Bever CA, Sauboin C, Galactionova K, Flasche S, et al. Public health impact and cost-effectiveness of the RTS, S/AS01 malaria vaccine: a systematic comparison of predictions from four mathematical models. Lancet Med. 2016;386:367–75.
WHO. Malaria policy advisory group (MPAG) meeting report, October 2021. Geneva: World Health Organization; 2021.
Download references
Acknowledgements
We acknowledge all participants for their engagement and for the additional questions raised during the masterclass. We also acknowledge the participants for reviewing the final manuscript and approving it for publication.
There was no funding for this work.
Author information
Authors and affiliations.
Johns Hopkins Center for Communication Programs, Baltimore, USA
April Monroe
MESA Alliance, Barcelona Institute for Global Health (ISGlobal), Barcelona, Spain
Nana Aba Williams
Barcelona Institute for Global Health (ISGlobal), Hospital Clínic-Universitat de Barcelona, Barcelona, Spain
Clinton Health Access Initiative, Boston, USA
Sheila Ogoma
Quality and Equity Healthcare, Kigali, Rwanda
Corine Karema
Swiss Tropical & Public Health Institute, Basel, Switzerland
Ifakara Health Institute, Ifakara, Tanzania
April Monroe & Fredros Okumu
You can also search for this author in PubMed Google Scholar
Contributions
AM and FO drafted the manuscript. NAW, SO and CK reviewed and contributed to the draft. All authors read and approved the final manuscript.
Corresponding author
Correspondence to Fredros Okumu .
Ethics declarations
Competing interests.
The authors declare that they have no competing interests.
Additional information
Publisher's note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Monroe, A., Williams, N.A., Ogoma, S. et al. Reflections on the 2021 World Malaria Report and the future of malaria control. Malar J 21 , 154 (2022). https://doi.org/10.1186/s12936-022-04178-7
Download citation
Published : 27 May 2022
DOI : https://doi.org/10.1186/s12936-022-04178-7
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- World Malaria Report
Malaria Journal
ISSN: 1475-2875
- Submission enquiries: [email protected]
- Ecology of Malaria
- Human Factors and Malaria
- Sickle Cell
The natural history of malaria involves cyclical infection of humans and female Anopheles mosquitoes. In humans, the parasites grow and multiply first in the liver cells and then in the red cells of the blood. In the blood, successive broods of parasites grow inside the red cells and destroy them, releasing daughter parasites (“merozoites”) that continue the cycle by invading other red cells.
The blood stage parasites are those that cause the symptoms of malaria. When certain forms of blood stage parasites (gametocytes, which occur in male and female forms) are ingested during blood feeding by a female Anopheles mosquito, they mate in the gut of the mosquito and begin a cycle of growth and multiplication in the mosquito. After 10-18 days, a form of the parasite called a sporozoite migrates to the mosquito’s salivary glands. When the Anopheles mosquito takes a blood meal on another human, anticoagulant saliva is injected together with the sporozoites, which migrate to the liver, thereby beginning a new cycle.
Thus the infected mosquito carries the disease from one human to another (acting as a “vector”), while infected humans transmit the parasite to the mosquito, In contrast to the human host, the mosquito vector does not suffer from the presence of the parasites.
More on: Anopheles Mosquitoes
More on: Malaria Parasites
Human Factors And Malaria
Biologic characteristics and behavioral traits can influence an individual’s risk of developing malaria and, on a larger scale, the intensity of transmission in a population.
More on: Human Factors and Malaria
Where does malaria transmission occur?
For malaria transmission to occur, conditions must be such so that all three components of the malaria life cycle are present:
- Anopheles mosquitoes, which able to feed on humans humans, and in which the parasites can complete the “invertebrate host” half of their life cycle
- Humans. who can be bitten by Anopheles mosquitoes, and in whom the parasites can complete the “vertebrate host” half of their life cycle
- Malaria parasites.
In rare cases malaria parasites can be transmitted from one person to another without requiring passage through a mosquito (from mother to child in "congenital malaria" or through transfusion, organ transplantation, or shared needles.)
Climate is a key determinant of both the geographic distribution and the seasonality of malaria. Without sufficient rainfall, mosquitoes cannot survive, and if not sufficiently warm, parasites cannot survive in the mosquito.
Anopheles lay their eggs in a variety of fresh or brackish bodies of water, with different species having different preferences. Eggs hatch within a few days, with resulting larvae spending 9-12 days to develop into adults in tropical areas. If larval habitats dry up before the process is completed, the larvae die; if rains are excessive, they may be flushed and destroyed. Life is precarious for mosquito larvae, with most perishing before becoming adults.
Life is usually short for adult mosquitoes as well, with temperature and humidity affecting longevity. Only older females can transmit malaria, as they must live long enough for sporozoites to develop and move to the salivary glands. This process takes a minimum of nine days when temperatures are warm (30°C or 86°F) and will take much longer at cooler temperatures. If temperatures are too cool (15°C or 59°F for Plasmodium vivax , 20°C or 68°F for P. falciparum ), development cannot be completed and malaria cannot be transmitted. Thus, malaria transmission is much more intense in warm and humid areas, with transmission possible in temperate areas only during summer months.
In warm climates people are more likely to sleep unprotected outdoors, thereby increasing exposure to night-biting Anopheles mosquitoes. During harvest seasons, agricultural workers might sleep in the fields or nearby locales, without protection against mosquito bites.
Anopheles Mosquitoes
The types (species) of Anopheles present in an area at a given time will influence the intensity of malaria transmission. Not all Anopheles are equally efficient vectors for transmitting malaria from one person to another. Those species that are most prone to bite humans are the most dangerous, as bites inflicted on animals that cannot be infected with human malaria break the chain of transmission. If the mosquito regularly bites humans, the chain of transmission is unbroken and more people will become infected. Some species are biologically unable to sustain development of human malaria parasites, while others are readily infected and produce large numbers of sporozoites (the parasite stage that is infective to humans).
Many of the most dangerous species bite human indoors. For these species insecticide treated mosquito nets and indoor residual spray (whereby the inner walls of dwellings are coated with a long-lasting insecticide) are effective interventions. Both of these interventions require attention to insecticide resistance, which will evolve if the same insecticide is used continuously in the same area.

Biologic characteristics (inborn and acquired) and behavioral traits can influence an individual’s malaria risk and, on a larger scale, the overall malaria ecology.
More on: Human Factors And Malaria
Characteristics of the malaria parasite can influence the occurrence of malaria and its impact on human populations, for example:
- Areas where P. falciparum predominates (such as Africa south of the Sahara) will suffer more disease and death than areas where other species, which tend to cause less severe manifestations, predominate
- P. vivax and P. ovale have stages (“hypnozoites”) that can remain dormant in the liver cells for extended periods of time (months to years) before reactivating and invading the blood. Such relapses can result in resumption of transmission after apparently successful control efforts, or can introduce malaria in an area that was malaria-free
- P. falciparum (and to a lesser extent P. vivax ) have developed strains that are resistant to antimalarial drugs. Such strains are not uniformly distributed. Constant monitoring of the susceptibility of these two parasite species to drugs used locally is critical to ensure effective treatment and successful control efforts. Travelers to malaria-risk areas should use for prevention only those drugs that will be protective in the areas to be visited.

Plasmodium falciparum predominates in Africa south of the Sahara, one reason why malaria is so severe in that area.
Animal Reservoirs
A certain species of malaria called P. knowlesi has recently been recognized to be a cause of significant numbers of human infections. P. knowlesi is a species that naturally infects macaques living in Southeast Asia. Humans living in close proximity to populations of these macaques may be at risk of infection with this zoonotic parasite.
Areas Where Malaria Is No Longer Endemic
Malaria transmission has been eliminated in many countries of the world, including the United States. However, in many of these countries (including the United States) Anopheles mosquitoes are still present. Also, cases of malaria still occur in non-endemic countries, mostly in returning travelers or immigrants (“imported malaria”). Thus the potential for reintroduction of active transmission of malaria exists in many non-endemic parts of the world. All patients must be diagnosed and treated promptly for their own benefit but also to prevent the reintroduction of malaria.
Genetic Factors
Biologic characteristics present from birth can protect against certain types of malaria. Two genetic factors, both associated with human red blood cells, have been shown to be epidemiologically important. Persons who have the sickle cell trait (heterozygotes for the abnormal hemoglobin gene HbS) are relatively protected against P. falciparum malaria and thus enjoy a biologic advantage. Because P. falciparum malaria has been a leading cause of death in Africa since remote times, the sickle cell trait is now more frequently found in Africa and in persons of African ancestry than in other population groups. In general, the prevalence of hemoglobin-related disorders and other blood cell dyscrasias, such as Hemoglobin C, the thalassemias and G6PD deficiency, are more prevalent in malaria endemic areas and are thought to provide protection from malarial disease.
Persons who are negative for the Duffy blood group have red blood cells that are resistant to infection by P. vivax . Since the majority of Africans are Duffy negative, P. vivax is rare in Africa south of the Sahara, especially West Africa. In that area, the niche of P. vivax has been taken over by P. ovale , a very similar parasite that does infect Duffy-negative persons.
Other genetic factors related to red blood cells also influence malaria, but to a lesser extent. Various genetic determinants (such as the “HLA complex,” which plays a role in control of immune responses) may equally influence an individual’s risk of developing severe malaria.
More on: Sickle Cell and Malaria
Acquired Immunity
Acquired immunity greatly influences how malaria affects an individual and a community. After repeated attacks of malaria a person may develop a partially protective immunity. Such “semi-immune” persons often can still be infected by malaria parasites but may not develop severe disease, and, in fact, frequently lack any typical malaria symptoms.
In areas with high P. falciparum transmission (most of Africa south of the Sahara), newborns will be protected during the first few months of life presumably by maternal antibodies transferred to them through the placenta. As these antibodies decrease with time, these young children become vulnerable to disease and death by malaria. If they survive repeated infections to an older age (2-5 years) they will have reached a protective semi-immune status. Thus in high transmission areas, young children are a major risk group and are targeted preferentially by malaria control interventions.
In areas with lower transmission (such as Asia and Latin America), infections are less frequent and a larger proportion of the older children and adults have no protective immunity. In such areas, malaria disease can be found in all age groups, and epidemics can occur.
Anemia in young children in Asembo Bay, a highly endemic area in western Kenya. Anemia occurs most between the ages of 6 and 24 months. After 24 months, it decreases because the children have built up their acquired immunity against malaria (and its consequence, anemia).

Mother and her newborn in Jabalpur Hospital, State of Madhya Pradesh, India. The mother had malaria, with infection of the placenta.
Pregnancy and Malaria
Pregnancy decreases immunity against many infectious diseases. Women who have developed protective immunity against P. falciparum tend to lose this protection when they become pregnant (especially during the first and second pregnancies). Malaria during pregnancy is harmful not only to the mothers but also to the unborn children. The latter are at greater risk of being delivered prematurely or with low birth weight, with consequently decreased chances of survival during the early months of life. For this reason pregnant women are also targeted (in addition to young children) for protection by malaria control programs in endemic countries.
More on: Malaria During Pregnancy
Behavioral Factors
Human behavior, often dictated by social and economic reasons, can influence the risk of malaria for individuals and communities. For example:
- Poor rural populations in malaria-endemic areas often cannot afford the housing and bed nets that would protect them from exposure to mosquitoes. These persons often lack the knowledge to recognize malaria and to treat it promptly and correctly. Often, cultural beliefs result in use of traditional, ineffective methods of treatment.
- Travelers from non-endemic areas may choose not to use insect repellent or medicines to prevent malaria. Reasons may include cost, inconvenience, or a lack of knowledge.
- Human activities can create breeding sites for larvae (standing water in irrigation ditches, burrow pits)
- Agricultural work such as harvesting (also influenced by climate) may force increased nighttime exposure to mosquito bites
- Raising domestic animals near the household may provide alternate sources of blood meals for Anopheles mosquitoes and thus decrease human exposure
- War, migrations (voluntary or forced) and tourism may expose non-immune individuals to an environment with high malaria transmission.
Human behavior in endemic countries also determines in part how successful malaria control activities will be in their efforts to decrease transmission. The governments of malaria-endemic countries often lack financial resources. As a consequence, health workers in the public sector are often underpaid and overworked. They lack equipment, drugs, training, and supervision. The local populations are aware of such situations when they occur, and cease relying on the public sector health facilities. Conversely, the private sector suffers from its own problems. Regulatory measures often do not exist or are not enforced. This encourages private consultations by unlicensed, costly health providers, and the anarchic prescription and sale of drugs (some of which are counterfeit products). Correcting this situation is a tremendous challenge that must be addressed if malaria control and ultimately elimination is to be successful.
Protective Effect of Sickle Cell Trait Against Malaria
The sickle cell gene is caused by a single amino acid mutation (valine instead of glutamate at the 6th position) in the beta chain of the hemoglobin gene. Inheritance of this mutated gene from both parents leads to sickle cell disease and people with this disease have shorter life expectancy. On the contrary, individuals who are carriers for the sickle cell disease (with one sickle gene and one normal hemoglobin gene, also known as sickle cell trait) have some protective advantage against malaria. As a result, the frequencies of sickle cell carriers are high in malaria-endemic areas.
CDC’s birth cohort studies (Asembo Bay Cohort Project in western Kenya) conducted in collaboration with the Kenya Medical Research Institute allowed an investigation into this issue. It was found that that the sickle cell trait provides 60% protection against overall mortality. Most of this protection occurs between 2-16 months of life, before the onset of clinical immunity in areas with intense transmission of malaria.
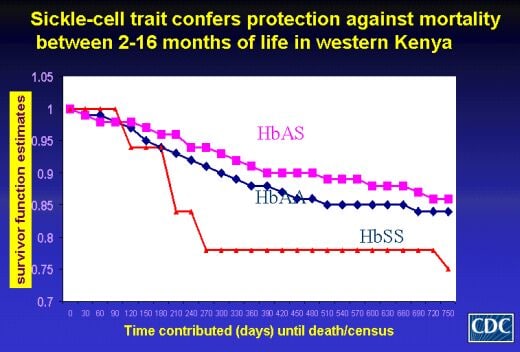
Graph of survival curves (“survival function estimates”) of children without any sickle cell genes (HbAA), children with sickle cell trait (HbAS), and children with sickle cell disease (HbSS). Those who had the sickle cell trait (HbAS) had a slight survival advantage over those without any sickle cell genes (HbAA), with children with sickle cell disease (HbSS) faring the worst.
Reference: Protective Effects of the Sickle Cell Gene Against Malaria Morbidity and Mortality. Aidoo M, Terlouw DJ, Kolczak MS, McElroy PD, ter Kuile FO, Kariuki S, Nahlen BL, Lal AA, Udhayakumar V. Lancet 2002; 359:1311-1312.
Malaria is transmitted to humans by female mosquitoes of the genus Anopheles. Female mosquitoes take blood meals for egg production, and these blood meals are the link between the human and the mosquito hosts in the parasite life cycle. The successful development of the malaria parasite in the mosquito (from the “gametocyte” stage to the “sporozoite” stage) depends on several factors. The most important is ambient temperature and humidity (higher temperatures accelerate the parasite growth in the mosquito) and whether the Anopheles survives long enough to allow the parasite to complete its cycle in the mosquito host (“sporogonic” or “extrinsic” cycle, duration 9 to 18 days). In contrast to the human host, the mosquito host does not suffer noticeably from the presence of the parasites.
Diagram of Adult Female Mosquito
Map of the world showing the distribution of predominant malaria vectors
Anopheles freeborni mosquito pumping blood Larger Picture
Sequential images of the mosquito taking its blood meal

General Information
There are approximately 3,500 species of mosquitoes grouped into 41 genera. Human malaria is transmitted only by females of the genus Anopheles . Of the approximately 430 Anopheles species, only 30-40 transmit malaria (i.e., are “vectors”) in nature. The rest either bite humans infrequently or cannot sustain development of malaria parasites.
Geographic Distribution
Anophelines are found worldwide except Antarctica. Malaria is transmitted by different Anopheles species in different geographic regions. Within geographic regions, different environments support a different species.
Anophelines that can transmit malaria are found not only in malaria-endemic areas, but also in areas where malaria has been eliminated. These areas are thus at risk of re-introduction of the disease.
Life Stages
Like all mosquitoes, anopheles mosquitoes go through four stages in their life cycle: egg, larva, pupa, and adult. The first three stages are aquatic and last 7-14 days, depending on the species and the ambient temperature. The biting female Anopheles mosquito may carry malaria. Male mosquitoes do not bite so cannot transmit malaria or other diseases. The adult females are generally short-lived, with only a small proportion living long enough (more than 10 days in tropical regions) to transmit malaria.
Adult females lay 50-200 eggs per oviposition. Eggs are laid singly directly on water and are unique in having floats on either side. Eggs are not resistant to drying and hatch within 2-3 days, although hatching may take up to 2-3 weeks in colder climates.
Mosquito larvae have a well-developed head with mouth brushes used for feeding, a large thorax, and a segmented abdomen. They have no legs. In contrast to other mosquitoes, Anopheles larvae lack a respiratory siphon and for this reason position themselves so that their body is parallel to the surface of the water.
Top: Anopheles Egg; note the lateral floats. Bottom: Anopheles eggs are laid singly.
Larvae breathe through spiracles located on the 8th abdominal segment and therefore must come to the surface frequently.
The larvae spend most of their time feeding on algae, bacteria, and other microorganisms in the surface microlayer. They do so by rotating their head 180 degrees and feeding from below the microlayer. Larvae dive below the surface only when disturbed. Larvae swim either by jerky movements of the entire body or through propulsion with the mouth brushes.
Larvae develop through 4 stages, or instars, after which they metamorphose into pupae. At the end of each instar, the larvae molt, shedding their exoskeleton, or skin, to allow for further growth.
Anopheles Larva. Note the position, parallel to the water surface.

The larvae occur in a wide range of habitats but most species prefer clean, unpolluted water. Larvae of Anopheles mosquitoes have been found in fresh- or salt-water marshes, mangrove swamps, rice fields, grassy ditches, the edges of streams and rivers, and small, temporary rain pools. Many species prefer habitats with vegetation. Others prefer habitats that have none. Some breed in open, sun-lit pools while others are found only in shaded breeding sites in forests. A few species breed in tree holes or the leaf axils of some plants.
The pupa is comma-shaped when viewed from the side. This is a transitional stage between larva and adult. The pupae does not feed, but undergoes radical metamorphosis. The head and thorax are merged into a cephalothorax with the abdomen curving around underneath. As with the larvae, pupae must come to the surface frequently to breathe, which they do through a pair of respiratory trumpets on the cephalothorax. After a few days as a pupa, the dorsal surface of the cephalothorax splits and the adult mosquito emerges onto the surface of the water.
The duration from egg to adult varies considerably among species and is strongly influenced by ambient temperature. Mosquitoes can develop from egg to adult in as little as 7 days but usually take 10-14 days in tropical conditions.
Anopheles Pupa
Anopheles Adults. Note (bottom row) the typical resting position.
Like all mosquitoes, adult anopheles have slender bodies with 3 sections: head, thorax and abdomen.
The head is specialized for acquiring sensory information and for feeding. The head contains the eyes and a pair of long, many-segmented antennae. The antennae are important for detecting host odors as well as odors of aquatic larval habitats where females lay eggs. The head also has an elongate, forward-projecting proboscis used for feeding, and two sensory palps.
The thorax is specialized for locomotion. Three pairs of legs and a single pair of wings are attached to the thorax.
The abdomen is specialized for food digestion and egg development. This segmented body part expands considerably when a female takes a blood meal. The blood is digested over time serving as a source of protein for the production of eggs, which gradually fill the abdomen.
Anopheles mosquitoes can be distinguished from other mosquitoes by the palps, which are as long as the proboscis, and by the presence of discrete blocks of black and white scales on the wings. Adult Anopheles can also be identified by their typical resting position: males and females rest with their abdomens sticking up in the air rather than parallel to the surface on which they are resting .
Adult mosquitoes usually mate within a few days after emerging from the pupal stage. In some species, the males form large swarms, usually around dusk, and the females fly into the swarms to mate. The mating habitats of many species remain unknown.
Males live for about a week, feeding on nectar and other sources of sugar. Females will also feed on sugar sources for energy but usually require a blood meal for the development of eggs. After obtaining a full blood meal, the female will rest for a few days while the blood is digested and eggs are developed. This process depends on the temperature but usually takes 2-3 days in tropical conditions. Once the eggs are fully developed, the female lays them then seeks blood to sustain another batch of eggs.
The cycle repeats itself until the female dies. Females can survive up to a month (or longer in captivity) but most do not live longer than 1-2 weeks in nature. Their chances of survival depend on temperature and humidity, but also upon their ability to successfully obtain a blood meal while avoiding host defenses.

Female Anopheles dirus feeding
Factors Involved in Malaria Transmission and Malaria Control
Understanding the biology and behavior of Anopheles mosquitoes can aid in designing appropriate control strategies. Factors that affect a mosquito’s ability to transmit malaria include its innate susceptibility to Plasmodium , its host choice, and its longevity. Long-lived species that prefer human blood and support parasite development are the most dangerous. Factors that should be taken into consideration when designing a control program include the susceptibility of malaria mosquitoes to insecticides and the preferred feeding and resting location of adult mosquitoes.
More on: How to Reduce Malaria’s Impact
Preferred Sources for Blood Meals
One important behavioral factor is the degree to which an Anopheles species prefers to feed on humans (anthropophily) or animals such as cattle (zoophily). Anthrophilic Anopheles are more likely to transmit the malaria parasites from one person to another. Most Anopheles mosquitoes are not exclusively anthropophilic or zoophilic; many are opportunistic and feed upon whatever host is available. However, the primary malaria vectors in Africa, An. gambiae and An. funestus , are strongly anthropophilic and, consequently, are two of the most efficient malaria vectors in the world.
Once ingested by a mosquito, malaria parasites must undergo development within the mosquito before they are infectious to humans. The time required for development in the mosquito (the extrinsic incubation period) takes 9 days or longer, depending on the parasite species and the temperature. If a mosquito does not survive longer than the extrinsic incubation period, then she will not be able to transmit any malaria parasites.
It is not possible to measure directly the life span of mosquitoes in nature, but many studies have indirectly measured longevity by examination of their reproductive status or via marking, releasing, and recapturing adult mosquitoes. The majority of mosquitoes do not live long enough to transmit malaria, but some may live as long as three weeks in nature. Though evidence suggests that mortality rate increases with age, most workers estimate longevity in terms of the probability that a mosquito will live one day. Usually these estimates range from a low of 0.7 to a high of 0.9. If survivorship is 90% daily, then a substantial proportion of the population would live longer than 2 weeks and would be capable of transmitting malaria. Any control measure that reduces the average lifespan of the mosquito population will reduce transmission potential. Insecticides thus need not kill the mosquitoes outright, but may be effective by limiting their lifespan.
Patterns of Feeding and Resting
Most Anopheles mosquitoes are crepuscular (active at dusk or dawn) or nocturnal (active at night). Some Anopheles mosquitoes feed indoors (endophagic) while others feed outdoors (exophagic). After blood feeding, some Anopheles mosquitoes prefer to rest indoors (endophilic) while others prefer to rest outdoors (exophilic). Biting by nocturnal, endophagic Anopheles mosquitoes can be markedly reduced through the use of insecticide-treated bed nets (ITNs) or through improved housing construction to prevent mosquito entry (e.g., window screens). Endophilic mosquitoes are readily controlled by indoor spraying of residual insecticides. In contrast, exophagic/exophilic vectors are best controlled through source reduction (destruction of larval habitats).
Insecticide Resistance
Insecticide-based control measures (e.g., indoor spraying with insecticides, ITNs) are the principal way to kill mosquitoes that bite indoors. However, after prolonged exposure to an insecticide over several generations, mosquitoes, like other insects, may develop resistance, a capacity to survive contact with an insecticide. Since mosquitoes can have many generations per year, high levels of resistance can arise very quickly. Resistance of mosquitoes to some insecticides has been documented within a few years after the insecticides were introduced. There are over 125 mosquito species with documented resistance to one or more insecticides. The development of resistance to insecticides used for indoor residual spraying was a major impediment during the Global Malaria Eradication Campaign. Judicious use of insecticides for mosquito control can limit the development and spread of resistance, particularly via rotation of different classes of insecticides used for control. Monitoring of resistance is essential to alert control programs to switch to more effective insecticides.
Susceptibility/Refractoriness
Some Anopheles species are poor vectors of malaria, as the parasites do not develop well (or at all) within them. There is also variation within species. In the laboratory, it has been possible to select for strains of An. gambiae that are refractory to infection by malaria parasites. These refractory strains have an immune response that encapsulates and kills the parasites after they have invaded the mosquito’s stomach wall. Scientists are studying the genetic mechanism for this response. It is hoped that some day, genetically modified mosquitoes that are refractory to malaria can replace wild mosquitoes, thereby limiting or eliminating malaria transmission.
Malaria Parasites
Malaria parasites are micro-organisms that belong to the genus Plasmodium . There are more than 100 species of Plasmodium , which can infect many animal species such as reptiles, birds, and various mammals. Four species of Plasmodium have long been recognized to infect humans in nature. In addition there is one species that naturally infects macaques which has recently been recognized to be a cause of zoonotic malaria in humans. (There are some additional species which can, exceptionally or under experimental conditions, infect humans.)

Ring-form trophozoites of P. falciparum in a thin blood smear.

Ring-form trophozoites of P. vivax in a thin blood smear.

Trophozoites of P. ovale in a thin blood smear.

Band-form trophozoites of P. malariae in a thin blood smear.

Schizont and ring-form trophozoite of P. knowlesi in a thin blood smear.
(All photos courtesy of DPDx )
The species infecting humans are the following:
- P. falciparum , which is found worldwide in tropical and subtropical areas, and especially in Africa where this species predominates. P. falciparum can cause severe malaria because it multiples rapidly in the blood, and can thus cause severe blood loss (anemia). In addition, the infected parasites can clog small blood vessels. When this occurs in the brain, cerebral malaria results, a complication that can be fatal.
- P. vivax , which is found mostly in Asia, Latin America, and in some parts of Africa. Because of the population densities especially in Asia it is probably the most prevalent human malaria parasite. P. vivax (as well as P. ovale ) has dormant liver stages (“hypnozoites”) that can activate and invade the blood (“relapse”) several months or years after the infecting mosquito bite.
- P. ovale is found mostly in Africa (especially West Africa) and the islands of the western Pacific. It is biologically and morphologically very similar to P. vivax . However, differently from P. vivax , it can infect individuals who are negative for the Duffy blood group, which is the case for many residents of sub-Saharan Africa. This explains the greater prevalence of P. ovale (rather than P. vivax ) in most of Africa.
- P. malariae , found worldwide, is the only human malaria parasite species that has a quartan cycle (three-day cycle). (The three other species have a tertian, two-day cycle.) If untreated, P. malariae causes a long-lasting, chronic infection that in some cases can last a lifetime. In some chronically infected patients P. malariae can cause serious complications such as the nephrotic syndrome.
- P. knowlesi is found throughout Southeast Asia as a natural pathogen of long-tailed and pig-tailed macaques. It has recently been shown to be a significant cause of zoonotic malaria in that region, particularly in Malaysia. P. knowlesi has a 24-hour replication cycle and so can rapidly progress from an uncomplicated to a severe infection; fatal cases have been reported.
- Disease caused by human malaria parasites.
- More images of the various stages of development of these 5 malaria species
To receive email updates about this page, enter your email address:
New! Locally Acquired Cases of Malaria in Florida, Texas, Maryland, and Arkansas
New! Update to Guidance for use of Artemether-Lumefantrine (Coartem®) in Pregnancy for Uncomplicated Malaria New! Discontinuation of CDC’s Distribution of Intravenous Artesunate as Commercial Drug Guidance for Malaria Diagnosis in Patients Suspected of Ebola Infection in the United States -->
See all Malaria Notices
- New! Malaria is a Serious Disease
- New! La malaria (paludismo) es una enfermedad grave
- How to Report a Case of Malaria
- CDC Yellow Book
- Red Pages: Malaria-endemic areas by country
- Drugs for Prevention
- Choosing a Drug to Prevent Malaria
- Drugs for Treatment in the U.S.
- Frequently Asked Questions (FAQs)
- Blood Banks
Click here for contact information
Exit Notification / Disclaimer Policy
- The Centers for Disease Control and Prevention (CDC) cannot attest to the accuracy of a non-federal website.
- Linking to a non-federal website does not constitute an endorsement by CDC or any of its employees of the sponsors or the information and products presented on the website.
- You will be subject to the destination website's privacy policy when you follow the link.
- CDC is not responsible for Section 508 compliance (accessibility) on other federal or private website.
Home / Essay Samples / Health / Illness / Malaria
Malaria Essay Examples
The impact of malaria disease on africa.
The “Villain in Africa”, otherwise known as Plasmodium or Malaria, is a parasite that affects nearly every continent. This horrific disease kills around one million to two million people each year. The majority of these victims are children in the Saharan parts of Africa. Mosquitos...
The Study Ofthe End of the Malaria-carrying Mosquitoes
Extinction of a species is often thought to be something negative and should be prevented at all costs, but what if we’re talking about the extinction of a “bad” species such as the malaria-carrying mosquitoes, Anopheles gambiae?Malaria is a disease caused by a parasite which...
Insecticides Resistance and Underlying Biological Mechanisms Which Aggravate the Burden of Malaria in Developing Countries
Despite considerable success of malaria control programme in the past, malaria still continues as a major public health problem. The malaria control relies mainly on indoor residual spraying of insecticides, which has become an inspiring fear task due to widespread resistance in malaria vectors, however,...
Crispr Technology as a Solution to Malaria Prevention
Malaria, a disease that is a transmitted person to person through an infected mosquito carrying the gene that allows for the parasite carrying the virus to exist. From a bar graph by Our World In Data, since 2015, about 395, 000 people have died in...
Youyou Tu – a Woman Who Made a Breakthrough in Malaria Treatment
The first women ever to win a Nobel prize in any discipline for China, a brilliant mind, and a crucial discovery in which would ultimately lead to lives saved and a brighter future where we can stand up to Malaria and fight it off. Youyou...
The Correlation Between Food Insecurity and Malaria in Haiti
Over half of Haiti’s population lives in extreme poverty, making it one of the most food insecure countries in the world. Vector-borne diseases that are being spread and transmitted through mosquitos are becoming a public health crisis across the globe. This is especially true in...
Mosquito Borne-diseases and Their Management
Mosquitoes are known as one of the fatal living organisms in the world acting as vectors (living organisms that can transmit infectious diseases between humans or from animals to humans) for different diseases. Their ability to transmit and disseminate disease to humans causes millions of...
Trying to find an excellent essay sample but no results?
Don’t waste your time and get a professional writer to help!
You may also like
- Assisted Suicide
- Nervous System
- Underage Drinking
- Organ Donation
- Digestive System
- Obesity Essays
- Hypertension Essays
- Diabetes Mellitus Essays
- Asthma Essays
- Cancer Essays
- Lung Cancer Essays
- Cervical Cancer Essays
- Hepatitis C Essays
- Drug Abuse Essays
samplius.com uses cookies to offer you the best service possible.By continuing we’ll assume you board with our cookie policy .--> -->